 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Founded in 1999, we are one of China's earliest and most trusted orthopedic implant R&D enterprises — delivering clinically proven solutions to patients and healthcare providers across 160+ countries and regions worldwide.
Founded in 1999, our company stands as one of the earliest enterprises in China dedicated to the research, development, and manufacturing of orthopedic implants. Over the past two and a half decades, we have grown from a pioneering domestic player into a globally recognized leader in the orthopedic medical device industry.
Our relentless commitment to R&D excellence, rigorous quality standards, and patient-centered design philosophy has enabled us to develop a comprehensive portfolio of orthopedic implant solutions — from joint reconstruction systems to spinal fixation devices and trauma implants — trusted by surgeons and healthcare institutions worldwide.
With sales channels spanning more than 160 countries and regions, and local regulatory approvals secured in over 60 markets, we combine global reach with deep local expertise. Our overseas offices in the United States, the Netherlands, and beyond ensure responsive support and seamless collaboration with our international partners.


What sets us apart in the global orthopedic implant industry — built on decades of expertise, innovation, and trust.
Our dedicated research and development team comprises over 200 engineers and clinical specialists. We invest significantly in innovation, holding hundreds of patents in orthopedic implant design, materials science, and minimally invasive surgical technologies.
Our ISO 13485-certified manufacturing facilities span over 50,000 square meters, equipped with precision CNC machining, cleanroom assembly environments, and automated quality inspection systems to ensure consistent, high-quality output at scale.
With local registrations secured in 60+ countries — including CE Mark (EU), FDA clearance (USA), and approvals across Asia-Pacific, Latin America, and the Middle East — we navigate complex regulatory landscapes seamlessly for our global partners.
Leveraging our agile manufacturing platform and robust supply chain, we deliver customized orthopedic implant solutions with shorter lead times than industry average — without compromising on quality or regulatory compliance.
Every product undergoes rigorous multi-stage quality control — from raw material verification to finished product testing — in compliance with ISO 13485, CE MDR, and other international medical device quality management standards.
Our international team provides comprehensive pre-sales consultation, regulatory guidance, clinical training, and after-sales technical support — ensuring our partners and end-users receive the highest level of service at every stage.
Thousands of healthcare providers and distributors worldwide trust us as their preferred orthopedic implant partner. Here's why.
One of China's first orthopedic implant R&D companies, with 25+ years of specialized experience and a proven track record of clinical excellence.
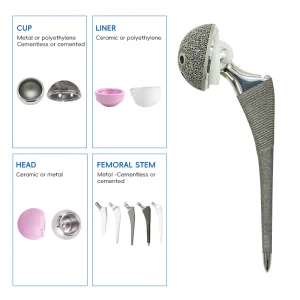
From joint replacement systems and spinal devices to trauma plates and screws — a one-stop orthopedic implant solution provider for all clinical needs.
Continuous investment in next-generation orthopedic technologies, including minimally invasive implant systems, bioresorbable materials, and smart implant solutions.
Physical presence in key markets with overseas offices in the USA and Netherlands, enabling local responsiveness backed by global manufacturing strength.
Our products meet the most stringent international medical device standards, giving our partners and patients complete confidence in quality and safety.
214 products certified under CE MDR, demonstrating full compliance with EU medical device regulations for safety and performance.
Our manufacturing and quality management system is fully certified to ISO 13485, the internationally recognized standard for medical device quality management.
Local regulatory approvals secured in more than 60 countries and regions, covering the Americas, Europe, Asia-Pacific, Africa, and the Middle East.
Products compliant with FDA requirements and major international regulatory frameworks, ensuring market access across North America and key global markets.
With over 100 global OEM partners, we are a trusted white-label and OEM manufacturer for leading medical device brands across North America, Europe, and Asia. Our OEM program is designed to deliver your branded orthopedic implants with the quality, regulatory compliance, and speed-to-market you demand.
Whether you need full product development from concept to finished device, or simply require high-volume precision manufacturing for your existing designs, our flexible OEM platform scales to meet your exact requirements — from prototype to millions of units annually.
A selection of successful collaborations and deployments that demonstrate our expertise and global impact in orthopedic implant solutions.
Delivered a comprehensive OEM manufacturing program for a leading US orthopedic brand, covering total hip and knee replacement systems. Full regulatory documentation support enabled successful FDA submission and market launch within 18 months.
Supplied CE-certified spinal pedicle screw systems and interbody fusion cages to a network of 40+ hospitals across Germany, France, and the Netherlands. Achieved zero product recall rate over 5 years of continuous supply.
Established a comprehensive trauma implant distribution network across 15 Asia-Pacific countries, including local registration support, clinical training programs, and dedicated technical service teams for distributor partners.
Trusted by distributors, OEM clients, and healthcare institutions across the globe — here's what they say about working with us.
"Working with this team has been transformative for our business. Their OEM capabilities are world-class, and the regulatory support they provided was instrumental in getting our products to market in the EU on time and on budget."
"The quality of their spinal implant systems is exceptional. We have been supplying their CE-certified products to our hospital network for over five years with zero quality complaints. A truly reliable long-term partner."
"Their team's depth of knowledge in orthopedic implant design and international regulatory affairs is unmatched. They helped us navigate registrations in 8 countries simultaneously — a feat we couldn't have achieved alone."
From our headquarters in China to offices in the USA and Netherlands, we maintain a truly global footprint to serve our international partners effectively.
Main R&D and manufacturing hub with 50,000+ m² facilities and 200+ engineers dedicated to orthopedic innovation.
North American operations center supporting FDA regulatory affairs, OEM client management, and US market distribution.
European hub managing CE regulatory compliance, EU distributor partnerships, and clinical support across the continent.
Active sales and distribution channels across the Americas, Europe, Asia-Pacific, Africa, and the Middle East.
Whether you're looking for OEM manufacturing, distribution partnerships, or regulatory support — our team is ready to help you succeed in the global orthopedic market.




When it comes to emergency situations, a Trauma Instrument Kit is essential. It serves as the first line of defense in treating injuries.
Author: Henry Date: 2026-03-30
Learn More
The 2026 Canton Fair will serve as a significant platform for showcasing advancements in medical technology, including the Pelvic External Fixator.
Author: Amelia Date: 2026-03-27
Learn More
The "Fixed Loop Endobutton" plays a crucial role in advancing medical technology, particularly in surgical settings. Its application in minimally
Author: Madeline Date: 2026-03-25
Learn More