 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Orthopedic Mini Instrument for Hand and Foot Surgery
Orthopedic Mini Instrument for Hand and Foot Surgery
 Orthopedic Instruments Femoral Nail Instrument Set
Orthopedic Instruments Femoral Nail Instrument Set
 Expert Femoral Instrument Set for Shoulder Surgery
Expert Femoral Instrument Set for Shoulder Surgery
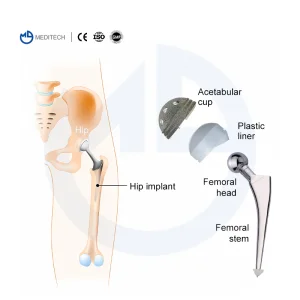 Cemented Stem Hip Replacement Implants
Cemented Stem Hip Replacement Implants
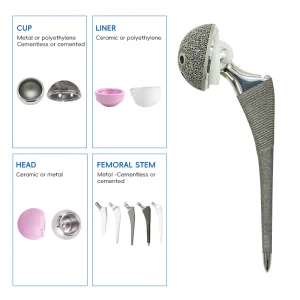 UHMWPE Insert Acetabular Cup System
UHMWPE Insert Acetabular Cup System
 CE Sterilized Prosthesis Knee Joint
CE Sterilized Prosthesis Knee Joint
 Knee Surgery & Artificial Hip Joint System
Knee Surgery & Artificial Hip Joint System
 Class III Knee Joint & Hip Prosthesis
Class III Knee Joint & Hip Prosthesis
The global orthopedic market is witnessing a significant shift toward Revision Hip and Knee Systems. As primary joint replacements become more common in younger, more active patients, the long-term demand for secondary or "revision" surgeries is skyrocketing. By 2030, revision knee replacements in the US alone are projected to increase by over 600%, while revision hip surgeries are expected to grow by nearly 140%.
Today, a Revision Hip/Knee Systems Manufacturer must not only provide implants but also comprehensive anatomical solutions. Factors such as bone loss, infection management (periprosthetic infections), and the need for modularity have turned this industry into a high-tech sector where material science meets robotic precision. Global suppliers are now prioritizing advanced biocompatible materials like titanium alloys and Ultra-High Molecular Weight Polyethylene (UHMWPE) to ensure longevity and stability in compromised bone environments.
The orthopedic industry is no longer just about "hardware." It is about digital integration and customized care. Key trends include:
Procurement departments in modern hospitals and medical distribution groups have evolved. They no longer look for the lowest price alone; they look for Total Value Ownership. This includes:
MDR (EU) and FDA (US) certifications are non-negotiable. Our 214 CE certificates ensure smooth entry into European markets.
Revision surgery requires specialized instrument sets. A manufacturer must provide a full ecosystem, from femoral nails to pelvic clamps.
Large distributors often require localized branding or specific modifications for local clinical habits.
The application of Revision Hip/Knee systems varies significantly by region:
In North America and Europe, the focus is on "Active Aging," where patients expect to return to sports after a revision. This requires highly durable UHMWPE inserts and advanced bearing surfaces.
In Emerging Markets (Asia, Latin America), the focus is often on treating complex trauma and neglected primary failures. Here, the versatility of systems like the Total Hip and Knee Joint Replacement Titanium Implant is crucial for dealing with varied bone qualities and larger anatomical variations.
Furthermore, the use of disposable systems, such as the Disposable Pulsed Lavage System, has become a standard in high-volume surgical centers to prevent cross-contamination and reduce theater turnover time.
As a leading Chinese manufacturer founded in 1999, we represent the pinnacle of orthopedic industrialization. Our advantages include:
From raw titanium smelting to final ETO sterilization, our vertical integration reduces costs and lead times. Unlike European or US brands that outsource production, we maintain 100% control over quality, ensuring every Hand and Foot Instrument Set or Pelvic C-Clamp meets rigorous standards.
With overseas offices in the United States and the Netherlands, we blend Western clinical insights with Eastern manufacturing efficiency. This hybrid approach allows us to develop products like the Expert Femoral Instrument Set that are both innovative and cost-effective.
Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas.
Our sales channels cover more than 160 countries and regions. We have obtained local registration in more than 60 countries and established more than 3 overseas offices in countries such as the United States and the Netherlands. With more than 100 global OEM partners and 214 products having obtained CE certification, we are a global leader in medical solutions.


 Sterilized Carton Package Knee Joint
Sterilized Carton Package Knee Joint
 Hip Joint Replacement Prosthesis Set
Hip Joint Replacement Prosthesis Set
 Revision Arthroplasty Titanium Hip Joint
Revision Arthroplasty Titanium Hip Joint
 Orthopedic Pulsed Lavage System
Orthopedic Pulsed Lavage System
 Hand and Foot Surgical Instrument Set
Hand and Foot Surgical Instrument Set
 Medical C Clamp Pelvic Instrument Set
Medical C Clamp Pelvic Instrument Set
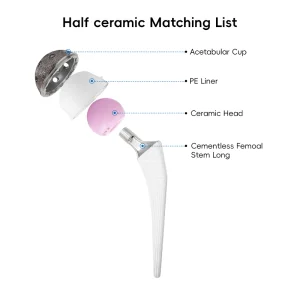 Artificial Hip and Knee Joint Replacement
Artificial Hip and Knee Joint Replacement
 Total Hip Titanium Implant Made in China
Total Hip Titanium Implant Made in China