




1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Customization: | Available |
|---|---|
| Customized: | Customized |
| Certification: | ISO, CE |




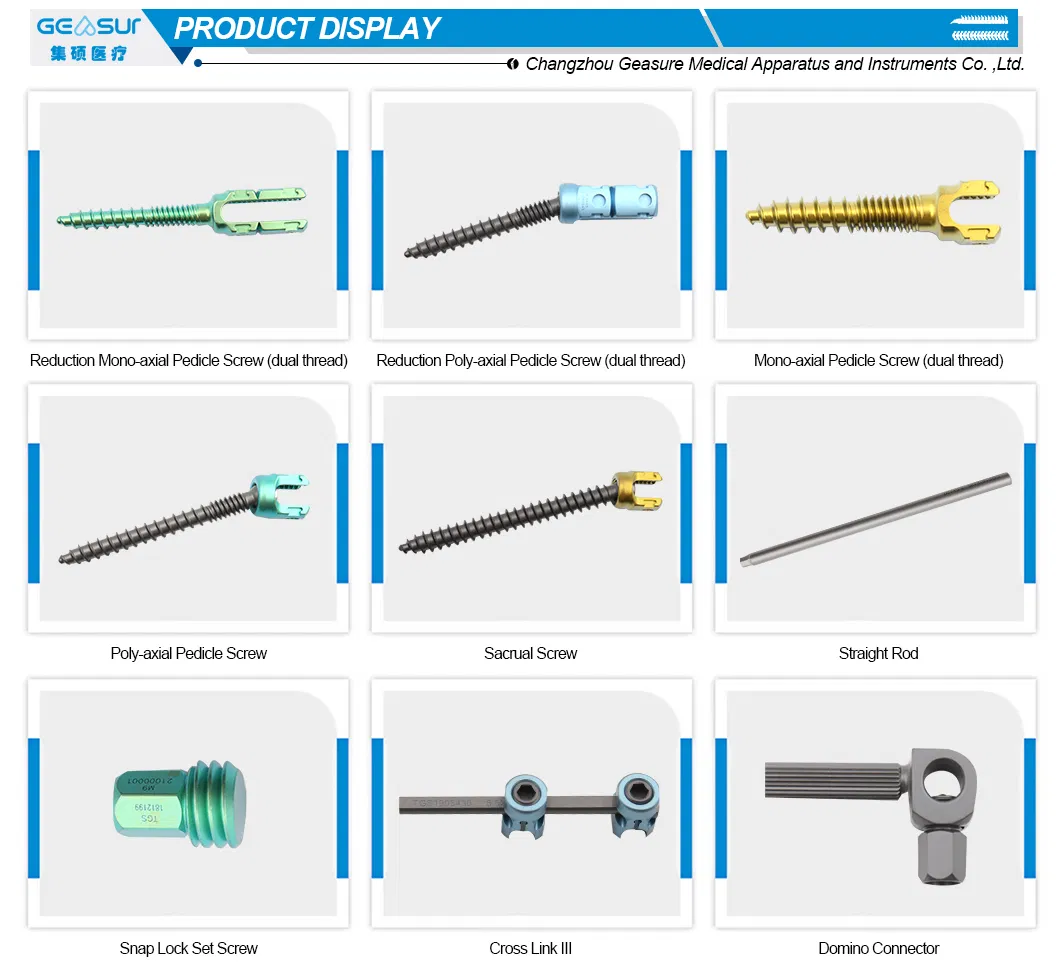
| Posterior Spinal Internal Fixation 5.5 System - Domino Connector | |||
|---|---|---|---|
| Cod (non sterile) | Cod (sterile) | Diameter (mm) | Length (mm) |
| 21005510 | 21085610 | 5.5 | 10 |
| 21005520 | 21085620 | 5.5 | 20 |
| 21005530 | 21085630 | 5.5 | 30 |

Established in October 2015, we are a professional entity specializing in the research and production of orthopedic implants and instruments, including trauma, spine, joint, and sports medicine. Our high-quality medical solutions serve the global orthopedic market.
Equipped with first-class production facilities and professional R&D teams, we maintain an advanced orthopedic laboratory. We collaborate with university professors and hospital surgeons to ensure our technology meets the highest clinical standards.
Our commitment to excellence is backed by ISO13485 and CE certifications. Many of our core products are registered with the FDA 510k, reflecting our dedication to quality and international compliance.



