




 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
Application: Widely used for different bone fracture of different bones.
Available Head: Hexagonal head
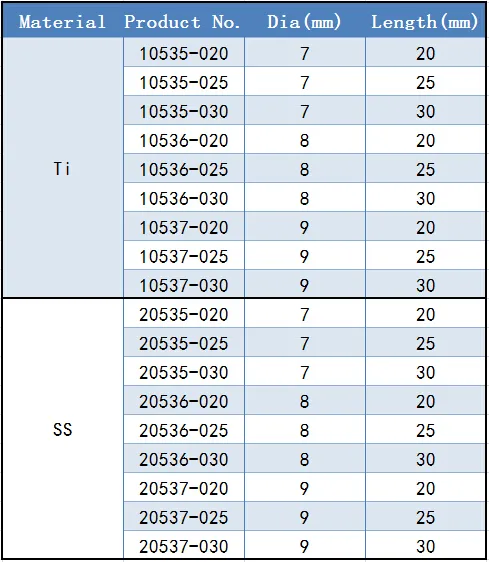
| Diameter | Length Options |
|---|---|
| 7.0mm | 20 / 25 / 30mm |
| 8.0mm | 20 / 25 / 30mm |
| 9.0mm | 20 / 25 / 30mm |
Instrument Set: Designed for use with ACL Interference Screw Instruments.


As a national high-tech enterprise, the proportion of R&D investment to total sales has exceeded 8% for consecutive years. This commitment realizes the transformation of clinical experience into innovative solutions, launching products with international invention patents.
Key revolutionary technologies include:
These systems are fully endorsed by global experts and serve as benchmarks for innovation in the medical device industry.
"Good faith as the base, quality above all"
The manufacturing process operates strictly according to the ISO13485 quality system, adhering to medical device manufacturing quality management, implant execution rules, and the CE MDD 93/42/EEC directive.
Our production facility features high-precision equipment including:
These resources ensure critical control over raw material procurement, product quality, and new product performance analysis.
Our orthopedic products are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Continuous feedback indicates that our product quality consistently meets and exceeds international standards.