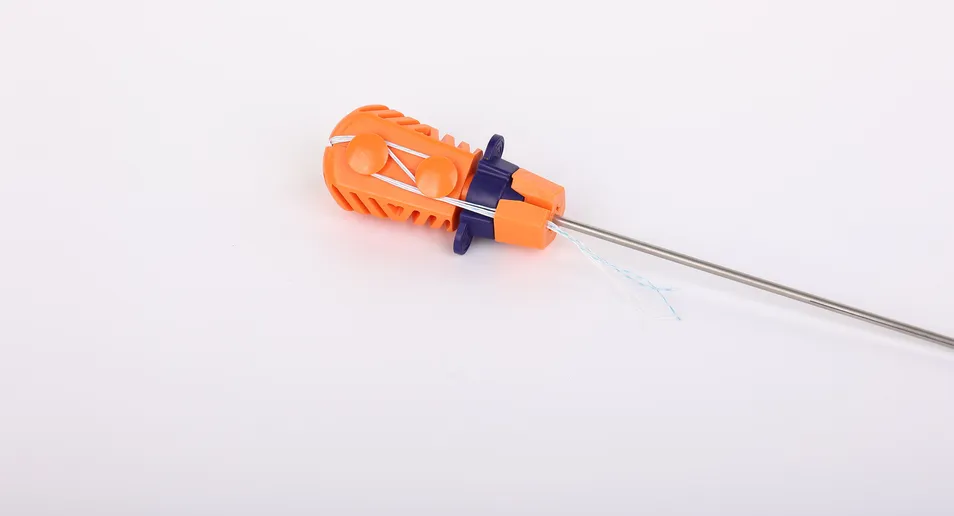
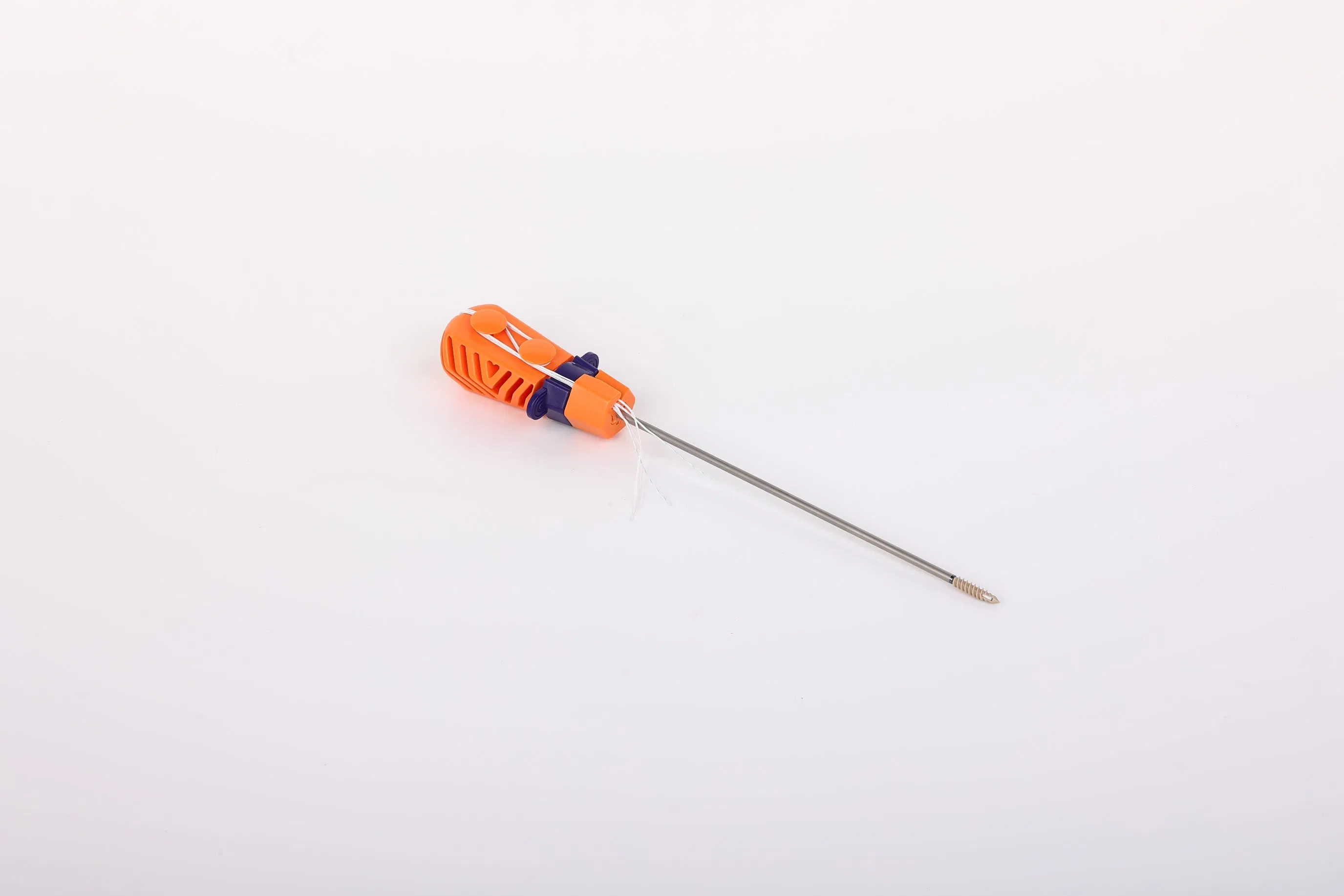
Suture Anchor System consists of anchor, nonabsorbable suture and inserter. Anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. Suture is made of ultra high molecular weight polyethylene material without coating. Blue suture is dyed with FDA approved material. The inserter is crafted from medical-grade stainless steel with an ergonomic handle.
Our comprehensive orthopedic range includes Disposable Surgical Lavage Systems, Suture Anchor Systems, Meniscal Repair Systems, Shaver Blades, Bone Cement Mixers, Rib Splints, and Finger Splints. To guarantee product quality and safety, manufacturing is conducted in Class 100,000 clean workshops and Class 10,000 purification laboratories. All products are fully compliant with CE certification and ISO 13485 standards.
?
Frequently Asked Questions
Are you a trading company or a manufacturer?
We are a professional manufacturer with our own specialized production facilities and an expert R&D team dedicated to orthopedic instruments.
Do you provide OEM services?
Yes, we offer comprehensive OEM services, including custom designs and tailored packaging solutions to align with your specific brand requirements.
Can I request a sample before placing a bulk order?
Yes, we provide samples for quality evaluation. Please contact our team to discuss the specific sample details and current lead times.
What certifications do your products hold?
All our surgical products are fully compliant with international quality benchmarks, holding CE certification and ISO 13485 standards.
What materials are used in the Suture Anchor System?
The anchors are made of high-strength Ti6Al4V alloy (ISO5832-3), and the sutures are made of ultra-high molecular weight polyethylene (UHMWPE) for maximum durability.
What is the warranty period for these orthopedic products?
We provide a 3-year warranty on our products, ensuring long-term reliability and support for medical institutions.



 Blutree Orthopedic Implants
Blutree Orthopedic Implants