

1 / 5
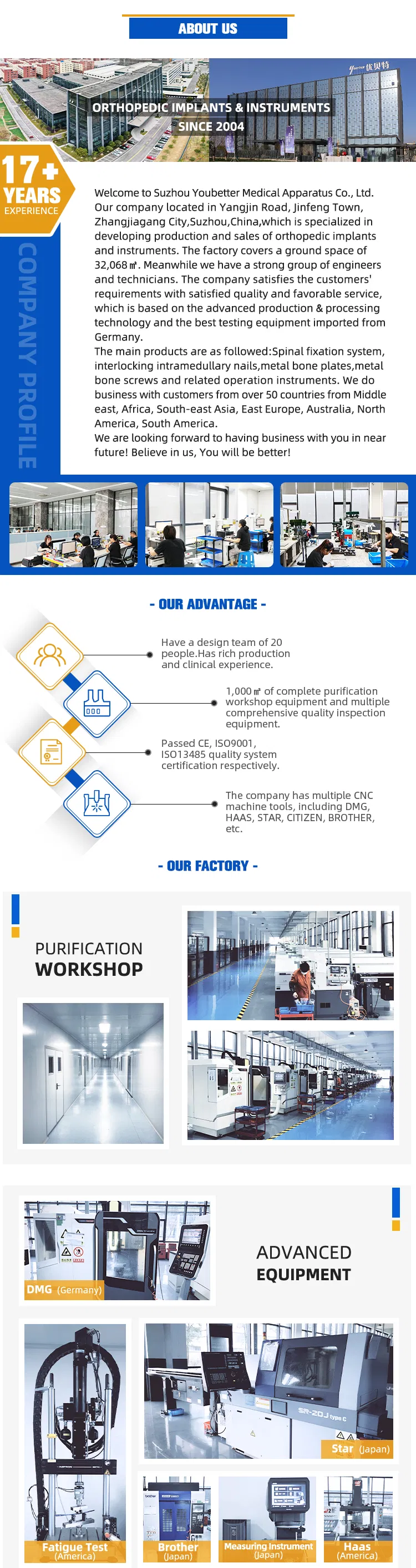
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |
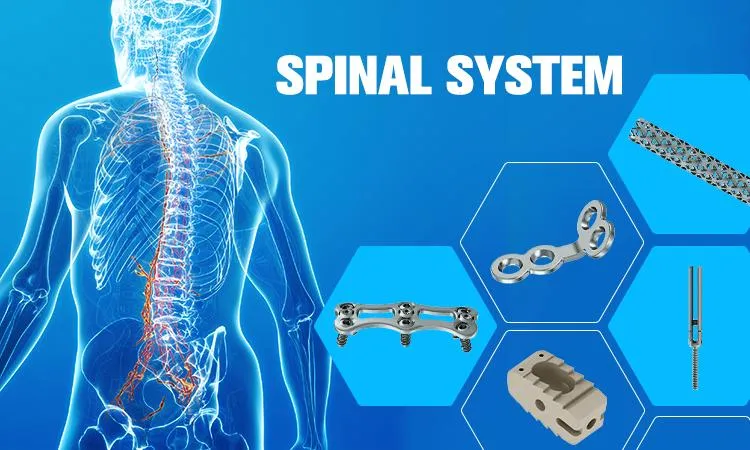

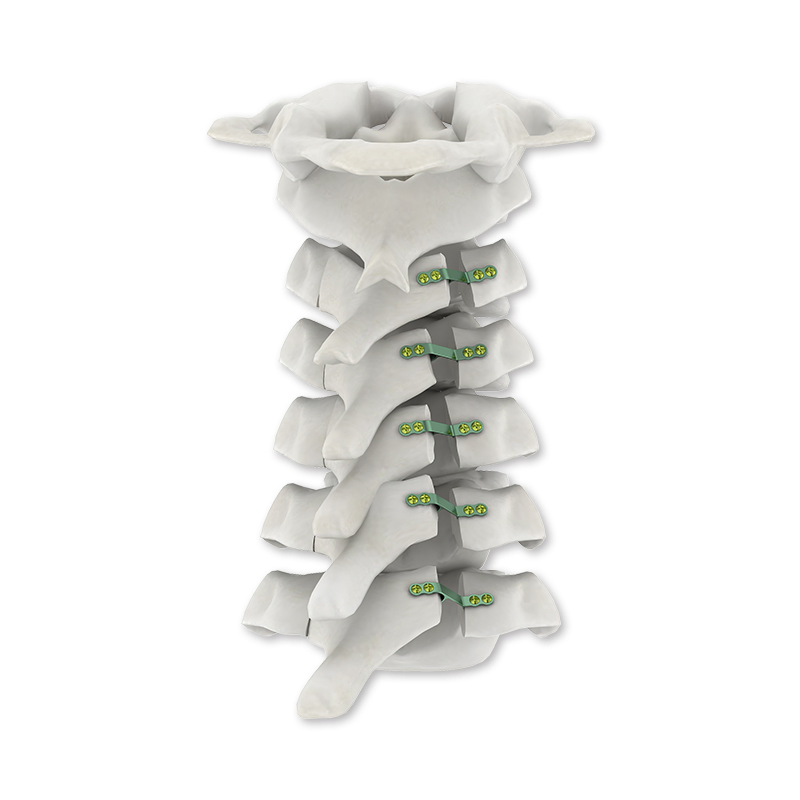
The mature implant system is designed to revolutionize spinal surgery. It provides reliable and efficient solutions for spinal internal fixation, available in both sterile or non-sterile packaging options for clinical convenience.


| Product Name | Spinal Internal Fixation 5.5/6.0 U-Multi-Axial Reduction Pedicle Screw |
| Material | Titanium |
| Color Options | Gold, Blue, Grey, Pink, etc. |
| Related Rod | 5.5 / 6.0 mm |
| Diameter | 4.5 / 5.0 / 5.5 / 6.0 / 6.5 / 7.0 mm |
| Length | 30 / 35 / 40 / 45 / 50 / 55 / 60 mm |
| Certificates | CE / ISO 13485 & 9001 |
| Package | Non-Sterilized / Sterilized |