




 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |


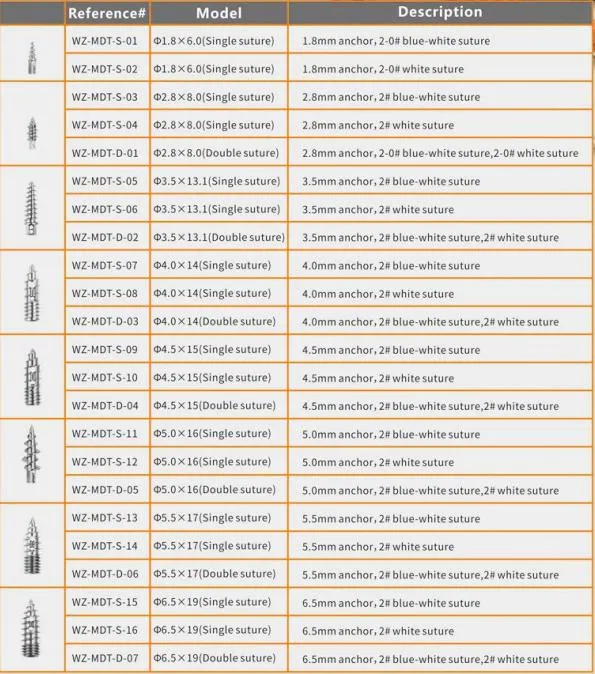
| Product Name | Non-bioabsorbable Suture Anchor |
| Carton Size | 42*22.5*25.5CM |
| G.W./CTN | 1.5KG - 1.8KG |
| Certificates | CE & ISO13485 |
| Period of Validity | 3 Years |
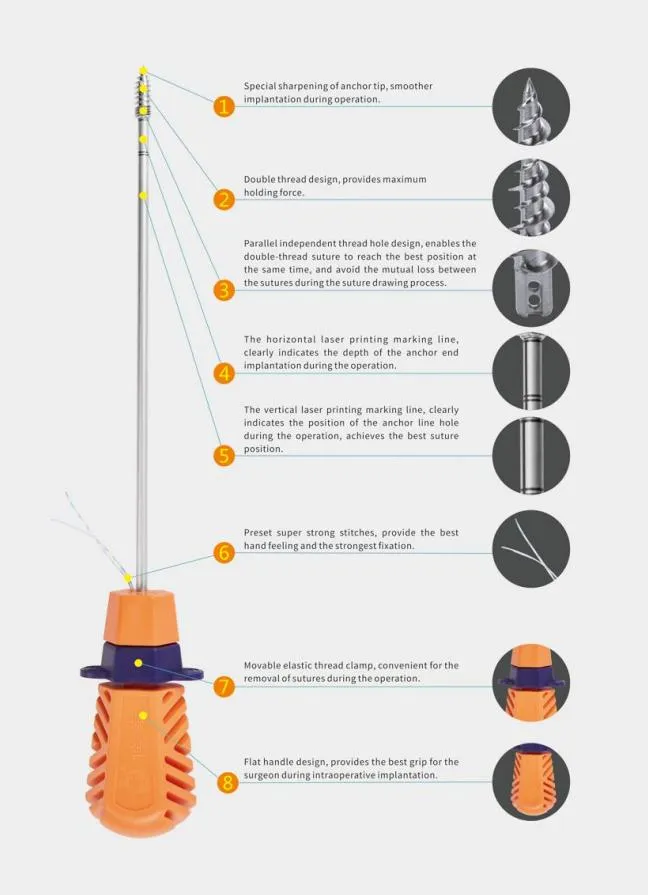
🚀 Different Head Options: Available in multiple configurations to suit various clinical requirements.
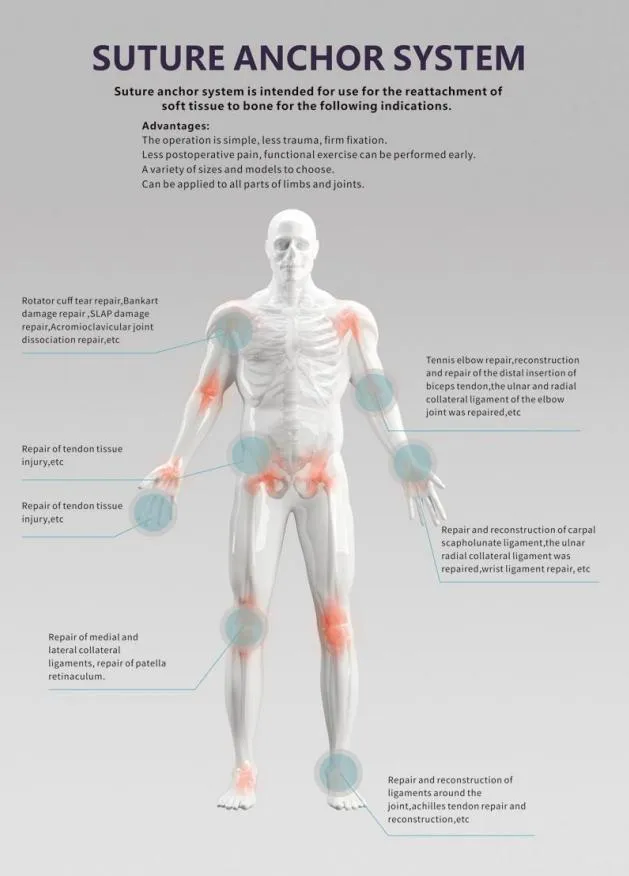


The manufacturing facility is a leading specialist in Disposable Surgical Systems designed for effective debridement following orthopedic procedures of trauma and joint arthroplasty. All products are certified under CE & ISO13485 standards, ensuring the highest quality for surgical applications.
The systems combine convenience and power for the removal of contaminated materials, necrotic tissue, and bone debris, providing controlled pressure for efficient irrigation at the wound site. Continuous improvement based on clinical research has led to optimized instruments with minimized noise and weight (approx. 772g), making them preferred tools for modern orthopedic surgeons.