Basic Information
Sterilization
Without Ethylene Oxide
Guarantee Period
Two Years
Customization
Available on Request
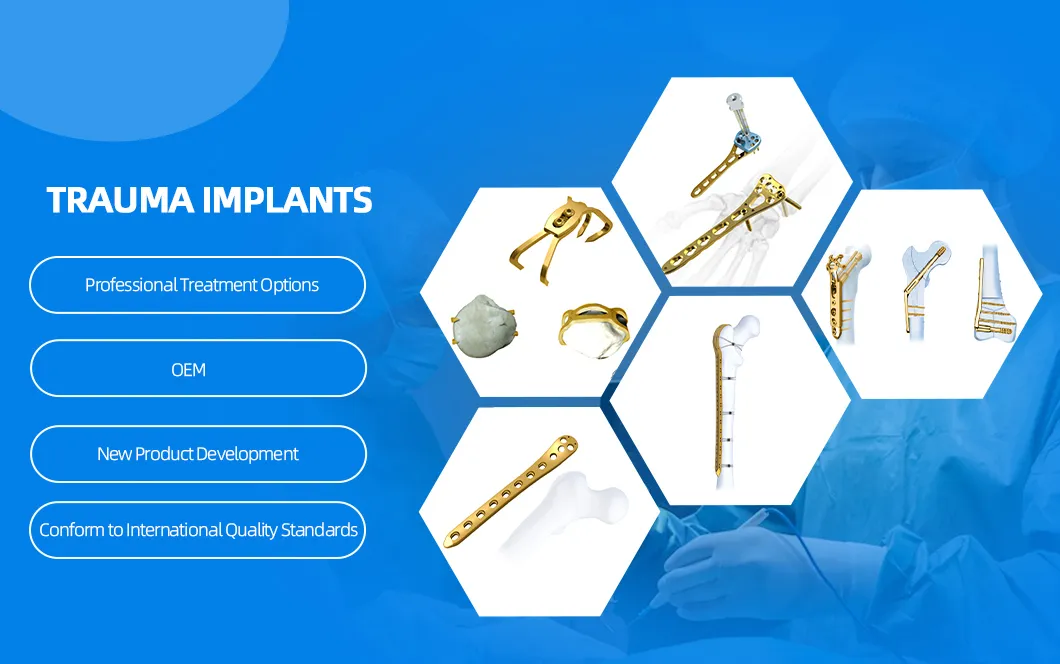
Usage
Fracture Fixation (Tibia)
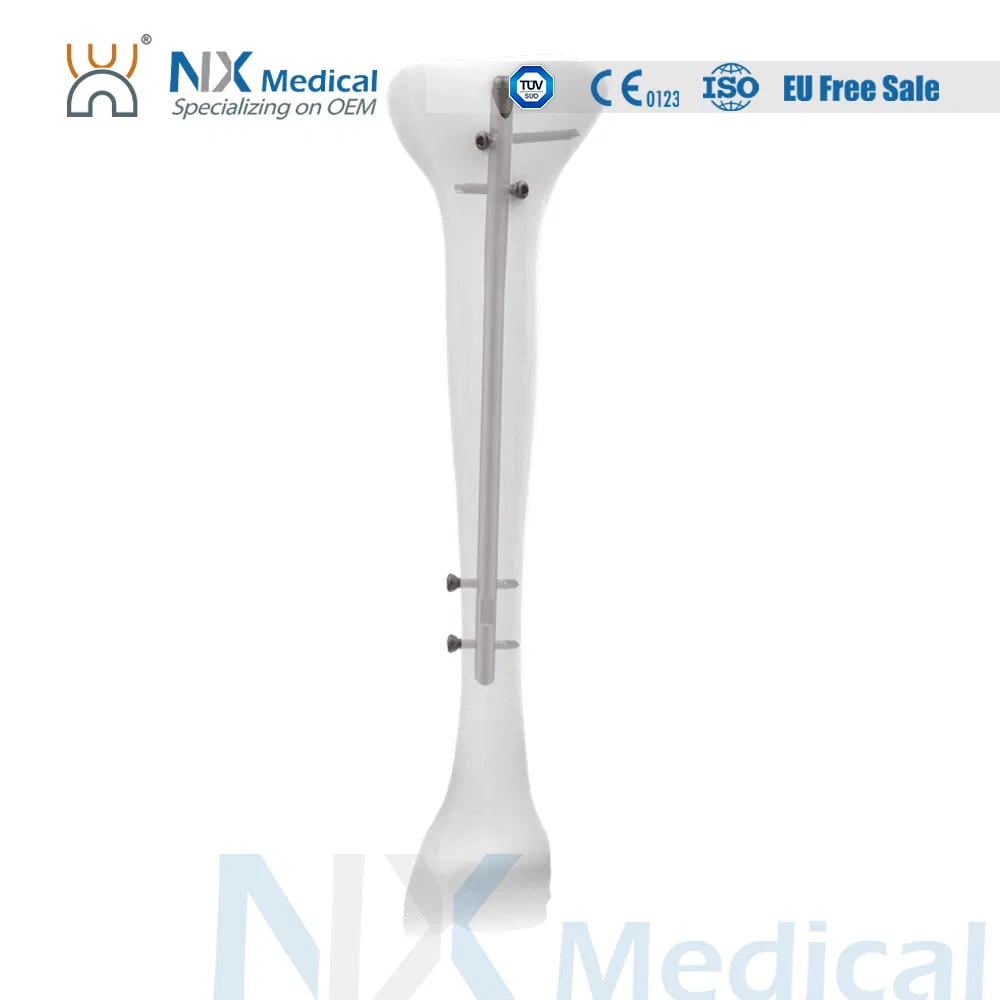
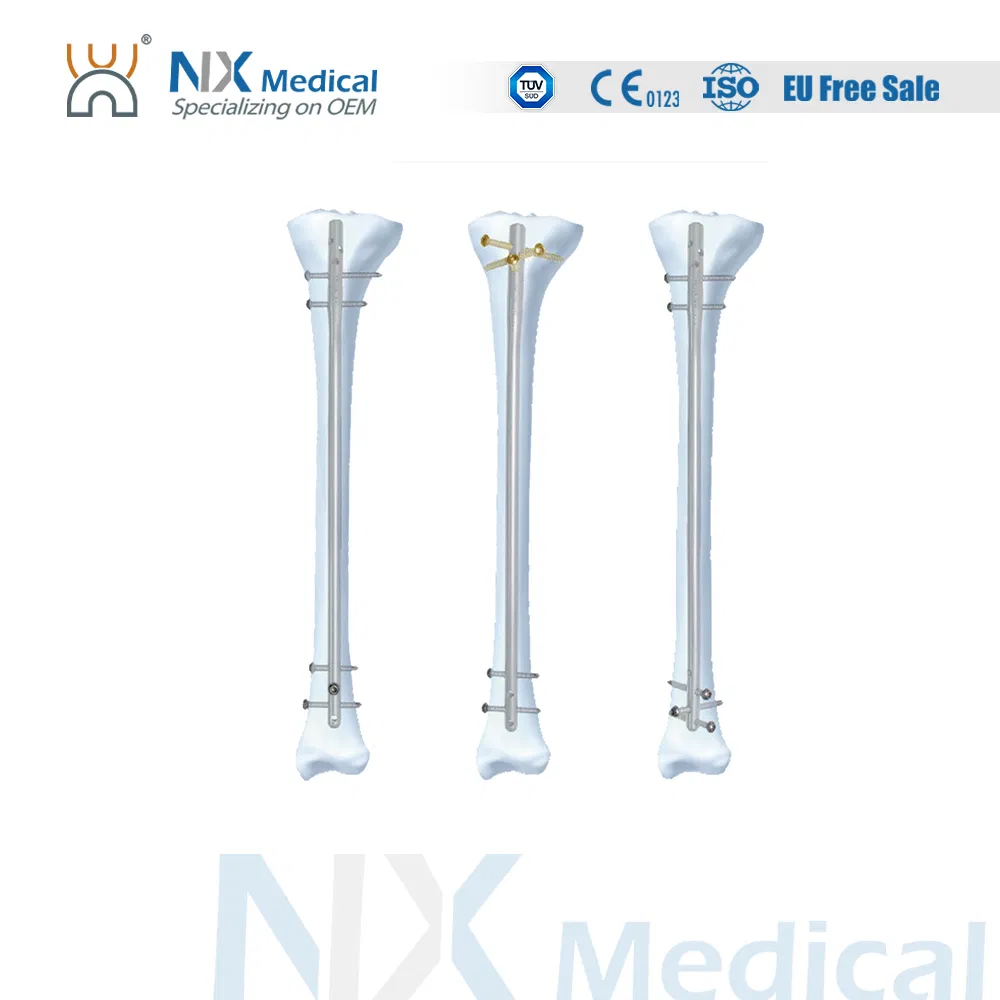
Product Description: Tibial Nail System
⚙️
Versatile Fixation: Different screws accommodate both cortical and cancellous bone fixation for optimal stability.
🎯
Easy Reduction: Elongated hole at the proximal side facilitates easier reduction during the surgical procedure.
📐
Advanced Insertion: Tapered nail tip design achieves easier nail insertion into the medullary canal.
🔗
Wider Treatment Range: Distal locking hole positioned close to the nail end tip allows for a wider range of fracture treatments.
🛠️
Precise Targeting: Dedicated targeting devices are available for every locking hole to ensure surgical accuracy.
Technical Specifications
| Product Name |
Diameter/Size (mm) |
Material |
| Tibial Nail |
φ8 / 9 / 10 / 11 (Length 255-420mm) |
Titanium Alloy (TA) |
| Interlocking Cancellous Screw |
φ4.8mm (Length 25-90mm) |
Titanium Alloy (TA) |
| Interlocking Screw |
φ4.4mm (Length 24-88mm) |
Titanium Alloy (TA) |
| Tibial Nail End Cap |
Extension: 0 / 5 / 10 / 15mm |
Titanium Alloy (TA) |
Company Profile
We are one of the leading Orthopedic product manufacturers located in East China, providing a comprehensive range of cost-effective surgical and reliable therapeutic solutions to orthopedic surgeons and patients. Our portfolio includes superior trauma implants, spinal implants, and professional surgical instruments.
Our major trauma categories feature locking plates, intramedullary nails, cannulated headless screws, and cable systems. For spinal systems, we offer cervical plates, pedicle screws, and PEEK cages. Quality is our priority; we have established a strict internal quality system and comply with ISO13485, with products certified by TUV CE0123 and NMPA.
Frequently Asked Questions
Can I request a sample order for quality testing?
Yes, we welcome sample orders to test and check the quality of our orthopedic implants. Mixed samples are also acceptable.
Do you offer customized manufacturing services?
Yes, we have a complete design and drawing department along with high-precision CNC machines to provide professional customized services.
What is the typical lead time for samples?
For existing items, samples are ready within 3 days. For OEM or customized products, the process generally takes approximately 2 weeks.
Can you provide branding with our company logo?
Yes, we can print your logo on both the products and the packaging, provided our minimum order quantity (MOQ) requirements are met.
How do you ensure the quality of the implants before shipment?
We conduct strict sampling inspections on all products before shipment and ensure that all packaging is intact and secure for transport.
How can we proceed with a formal order?
Firstly, let us know your requirements. We will then provide a quote. Once samples are confirmed and a deposit is placed, we will arrange production immediately.



 Blutree Orthopedic Implants
Blutree Orthopedic Implants