1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |

| Product Type | Holes | Direction | Material | Length (mm) |
|---|---|---|---|---|
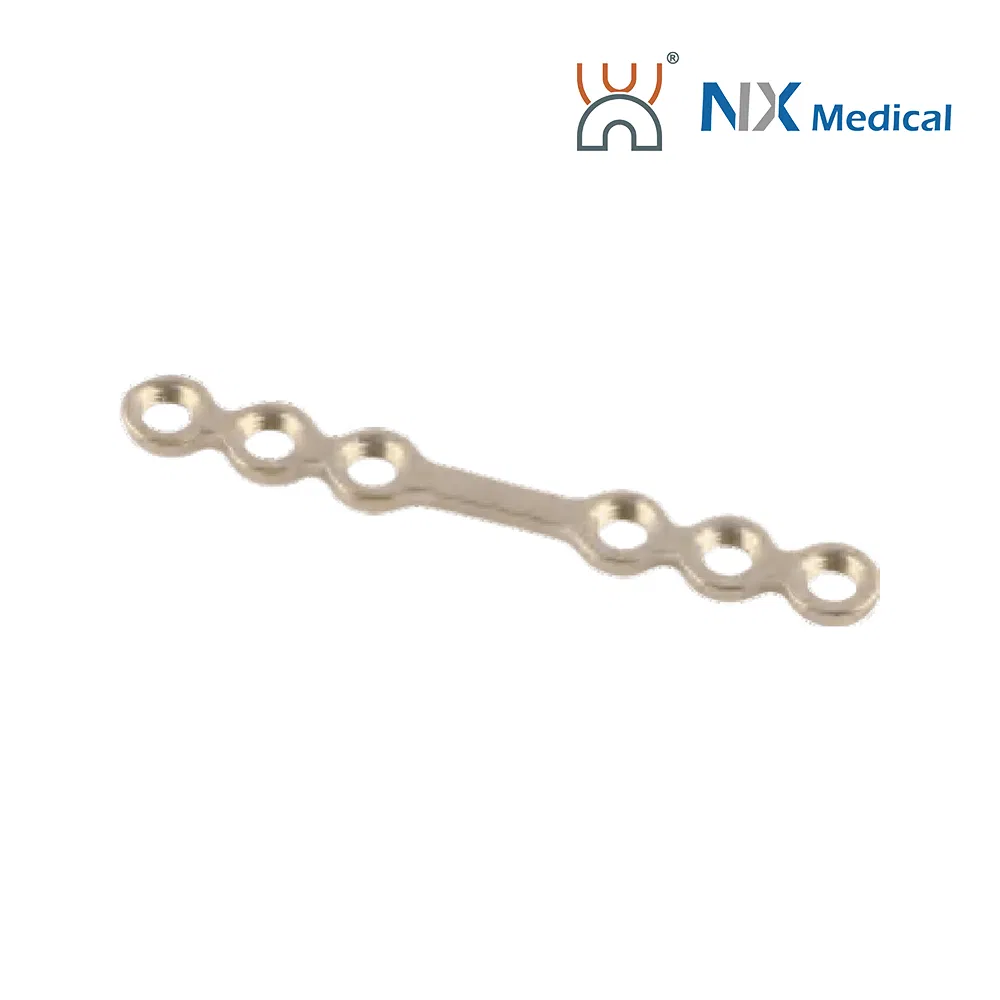
| 2.0mm Straight Bridge Repair LCP | 2 | - | Pure Titanium | 16.7 |
| 2.0mm Straight Bridge Repair LCP - II | 4H, 6-22mm | - | Pure Titanium | 20.7 - 36.7 |
| 2.0mm Arc Skull Reparing LCP | 4 - 10 | - | Pure Titanium | 22.5 - 52.6 |
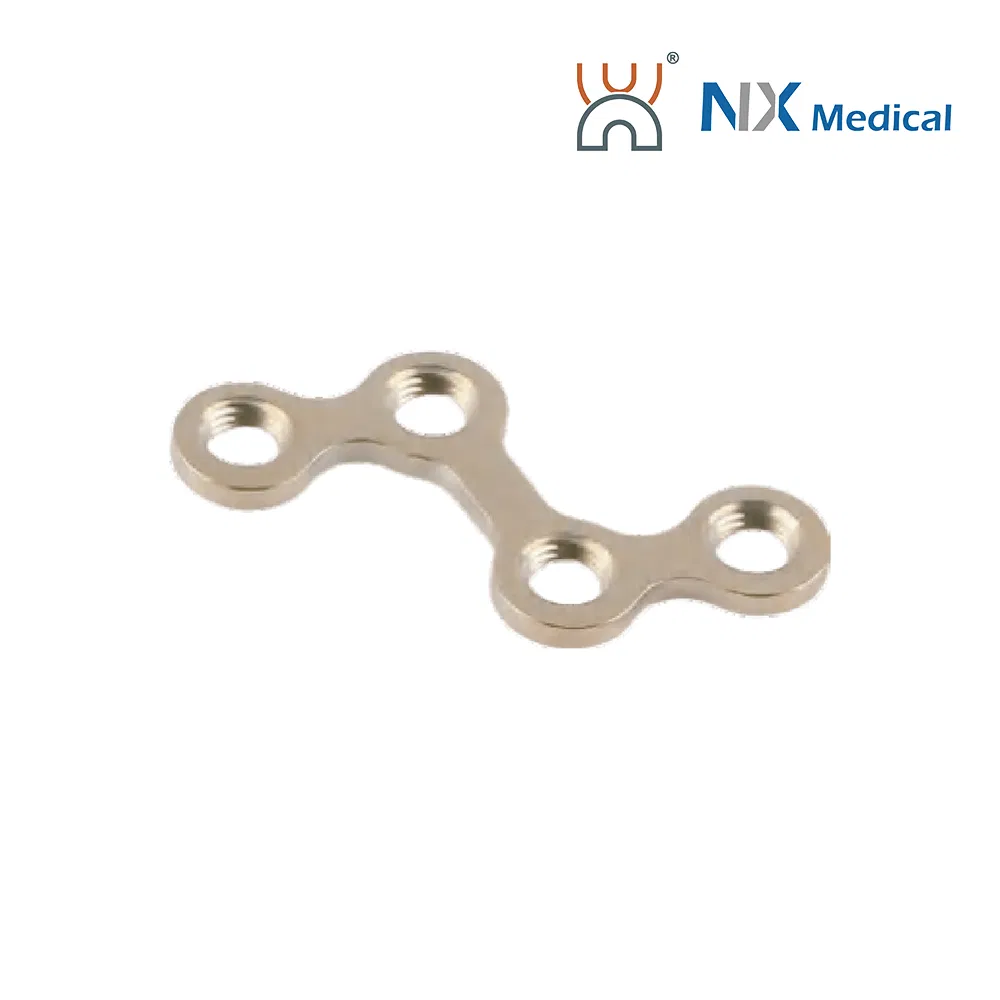
| 2.0mm T-type Cranioplasty LCP | 2+3H to 2+6H | - | Pure Titanium | 16.7 - 40.7 |
| 2.0mm Oblique T-type LCP | 2+4H | Left/Right | Pure Titanium | 29.2 |
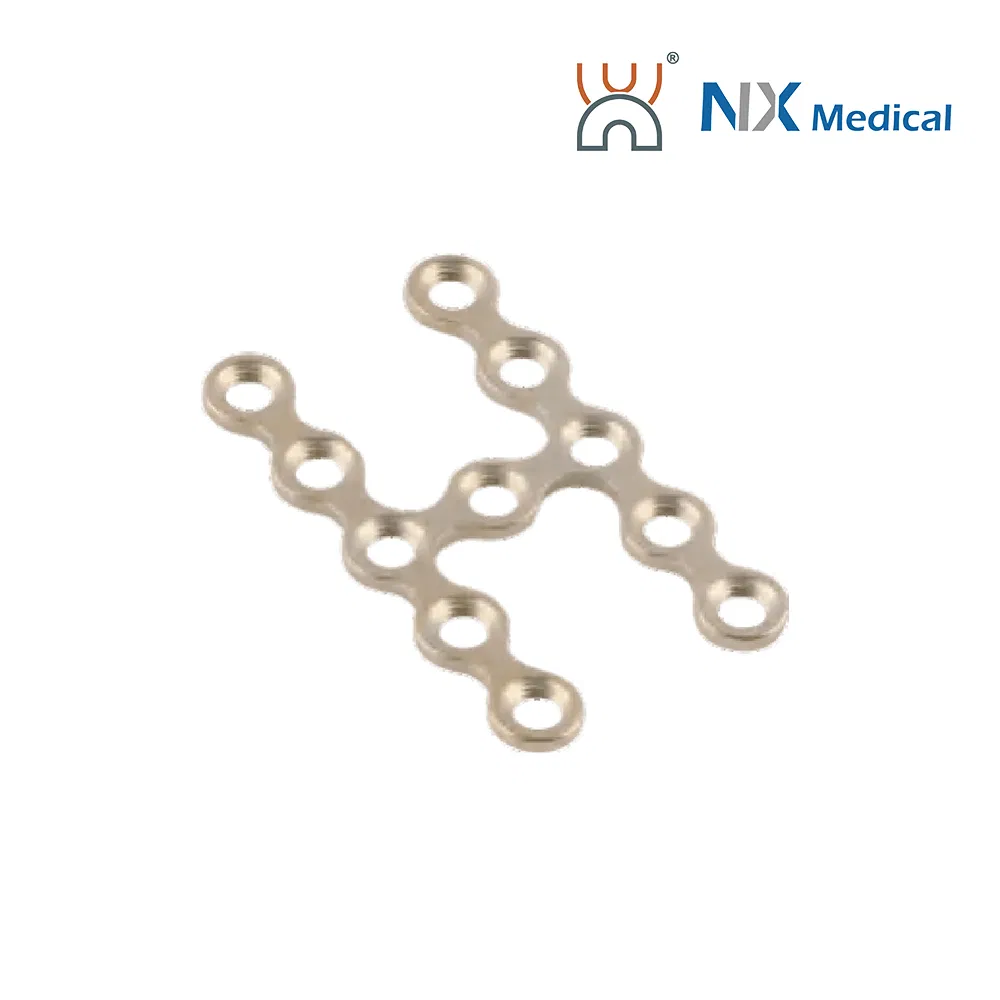
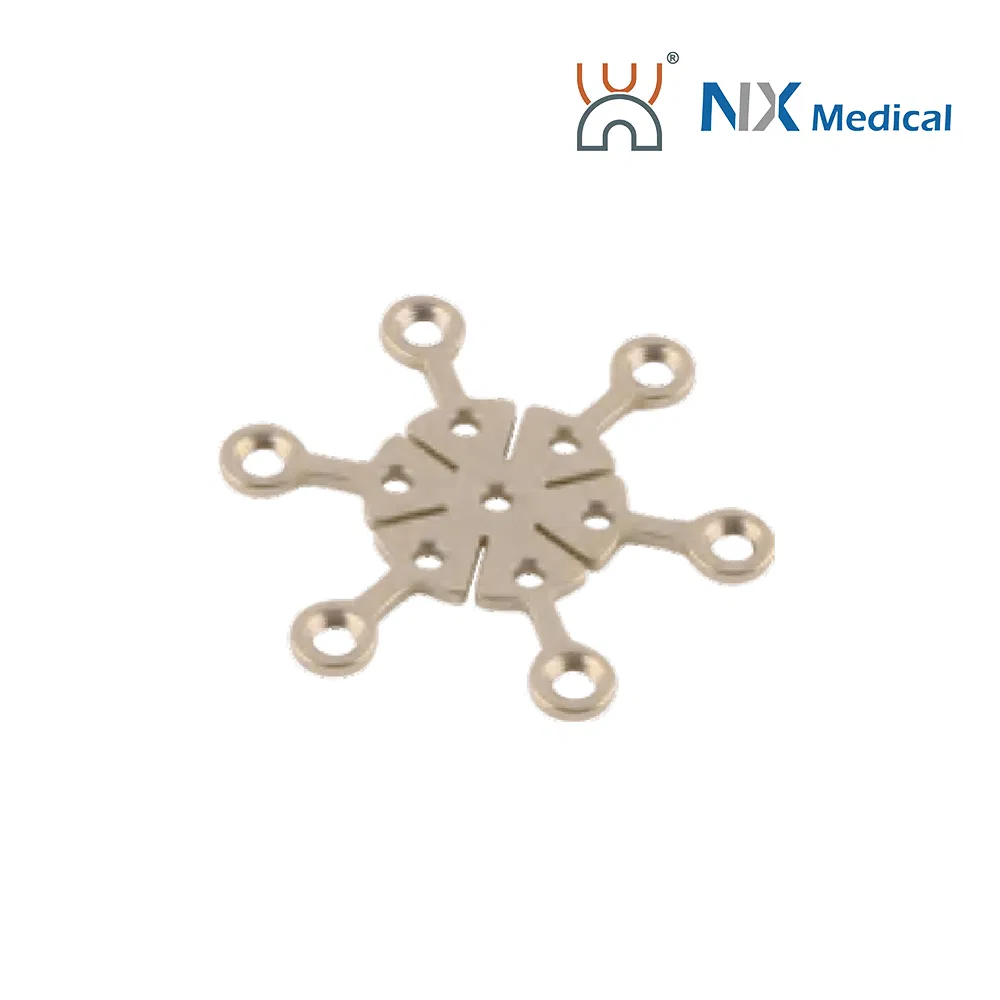
| 2.0mm X-type Cranioplasty LCP | 3+3H | - | Pure Titanium | 19.5 - 34.0 |
| 2.4mm Reconstruction LCP | 8+17H | - | Pure Titanium | 178.5 |

The manufacturer is a leading orthopedic product specialist located in East China, providing cost-effective surgical and reliable therapeutic solutions globally. We offer a comprehensive range of orthopedic trauma implants, spinal implants, and surgical instruments. Committed to continuous R&D, the company ensures that all medical devices meet the highest global standards.


Our major categories include locking plates, intramedullary nails, cannulated screws, and spinal systems such as cervical plates and pedicle screws. All products are manufactured under a strict quality system, complying with ISO13485, TUV CE0123, and NMPA certifications.