




1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 114/Piece Request Sample |
| Condition | New |
| Usage | Fracture Fixation |
| Using Site | Tibia Femur |
| Type | Trauma |
| Implant Type | Intramedullary Nail |
| Specification | Titanium Alloy TC4 |
| Origin | China |
Advantages of the Suprapatellar Approach:
Innovative Locking Mechanism with 5 holes in 4 directions. Dual-core threading increases screw strength.
Continuous long-curved contour reduces pressure on the medullary canal and matches Chinese anatomical structures.
Ultra-distal design with 4 holes in 3 directions ensures structural stability for complex fractures.

Indications:
Surgical Precautions:
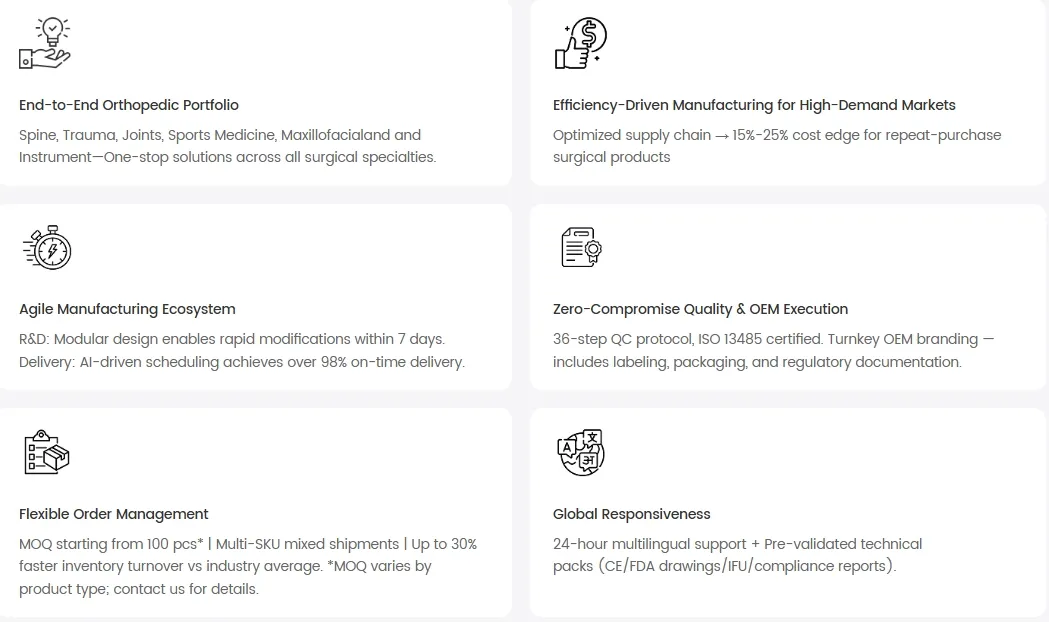
We are a leading manufacturer of standardized orthopedic implants and instruments, covering trauma, spine, joints, sports medicine, and CMF. Our facility includes in-house R&D, certified laboratories, and modern production lines.

