
1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
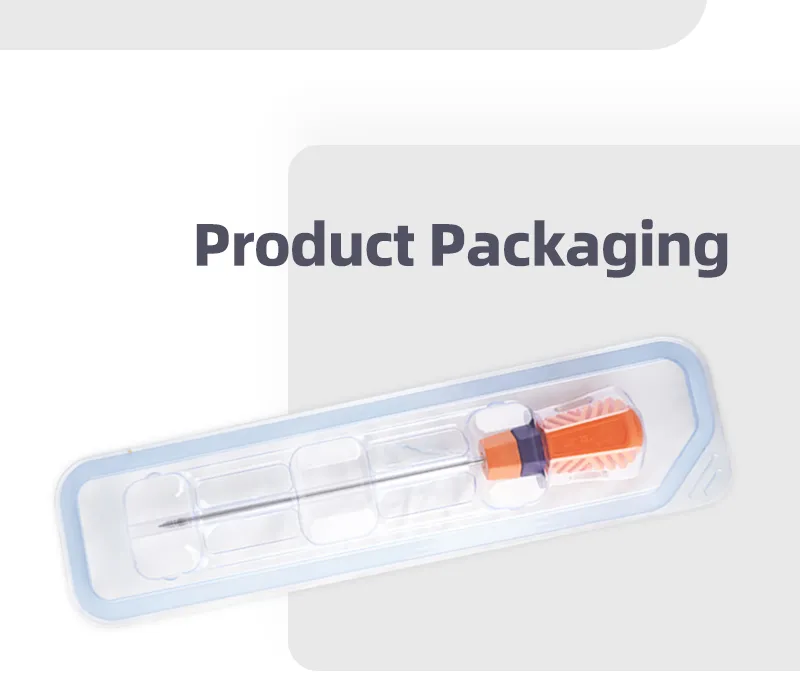
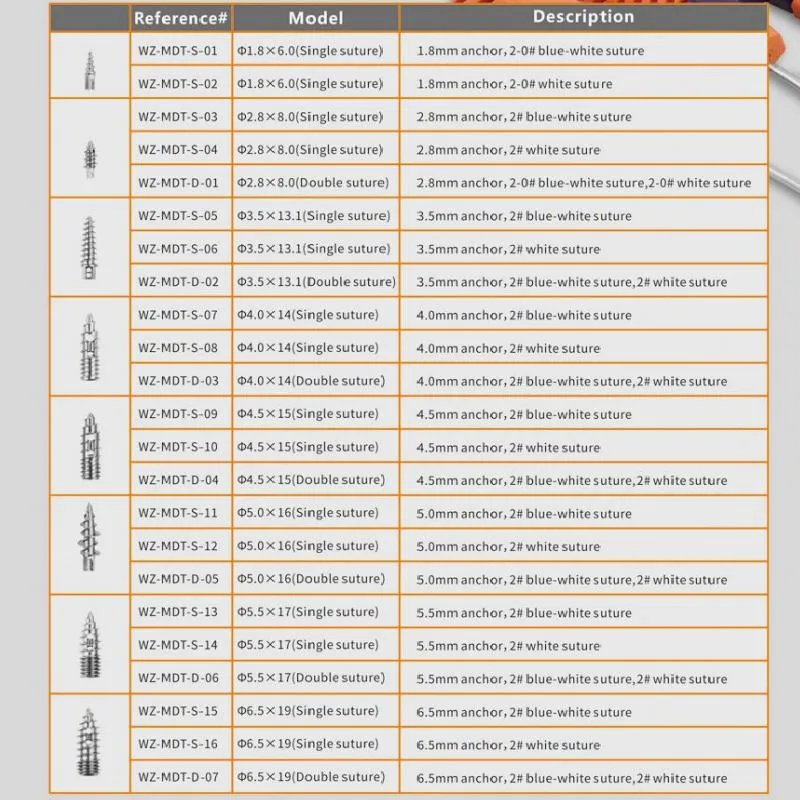
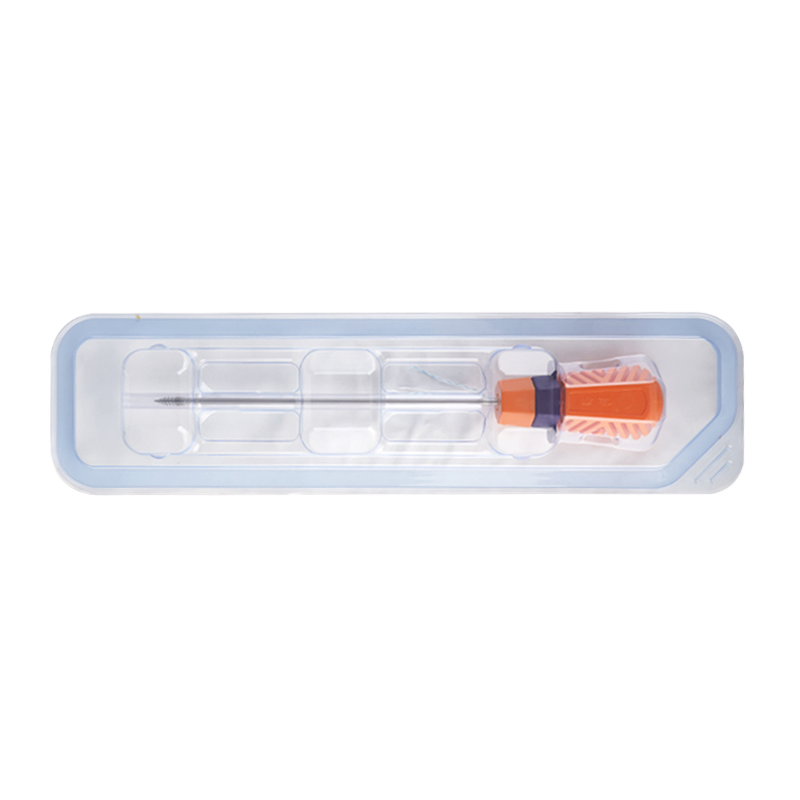
Suture Anchor Arthroscopy Implant for Knee Surgery Suture Anchor System
The Suture Anchor System consists of an anchor, nonabsorbable suture, and inserter. The anchor is manufactured from Ti6Al4V alloy, which strictly adheres to ISO5832-3:2016 requirements. The suture is crafted from ultra-high molecular weight polyethylene (UHMWPE) material without coating, woven from several UHMWPE yarns meeting ASTM F2848-17 standards.
The blue suture is derived from white UHMWPE dyed with blue material (code 73.1015), which is approved by the FDA. The part of the inserter that contacts the human body is made of high-quality stainless steel conforming to ASTM F899-12b, while the handle is constructed from durable polycarbonate and ABS material.