


 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 413/Piece Request Sample |
| Model NO. | 2410202 |
| Material | Titanium |
| Feature | Disposable |
| Certification | CE, FDA, ISO13485 |
| Group | Adult & Children |
| Product Name | Non-Absorbable Suture Anchor |
| MOQ | 1 Piece |
| OEM | Available |
| Package | PE Inner Bag + Carton |
| HS Code | 9021100090 |


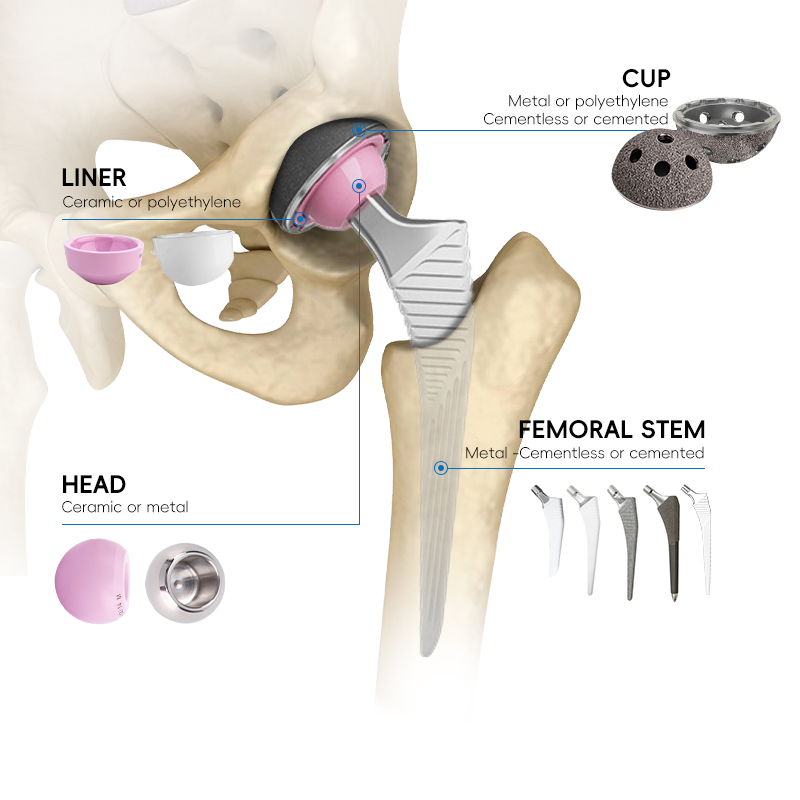
Modern implants utilize advanced bearing surfaces like ceramic-on-ceramic or ceramic-on-polyethylene, extending lifespan and reducing revision surgery risk.

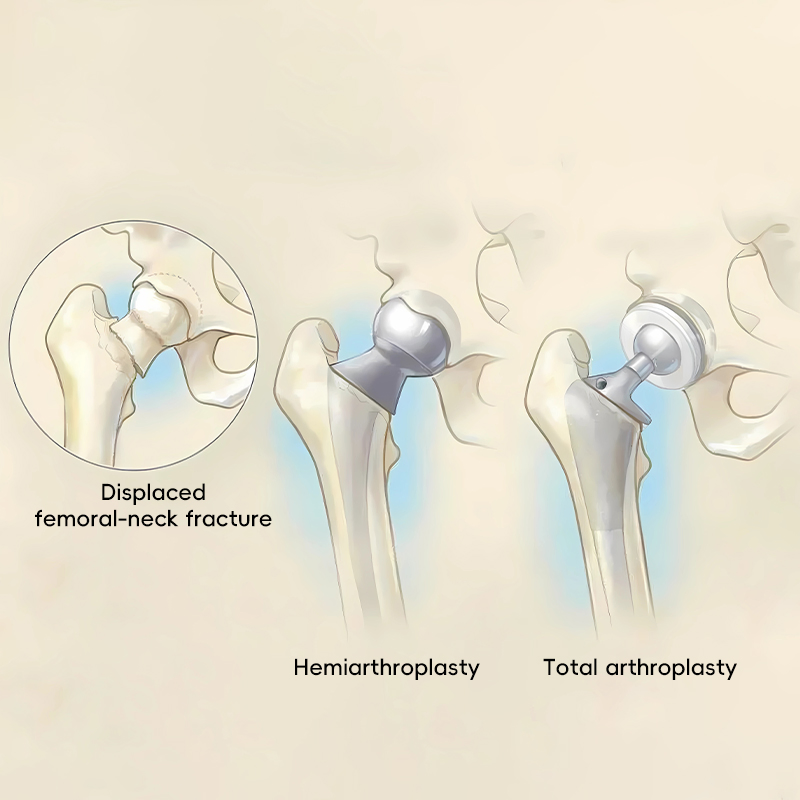
Covers both hemiarthroplasty (replacing only the femoral head) and total hip replacement (replacing both femoral head and acetabulum).
A modular system allows surgeons to tailor the procedure for each patient by assembling parts specific to their anatomy.

Each product has a clear matching list defining which femoral heads, stems, cups, and liners can be safely combined.

| Component | Introduction |
|---|---|
| Acetabular Cup | The "bowl" component implanted into the pelvis. Typically made of titanium or cobalt-chromium alloy with a porous surface for bone growth. |
| Acetabular Liner | A wear-resistant insert inside the cup. Serves as the bearing surface, made from polyethylene, ceramic, or metal to minimize friction. |
| Femoral Head | Spherical component that attaches to the stem neck. Rotates within the liner to enable range of motion. Made of ceramic or polished metal. |
| Femoral Stem | Inserted into the femur for stability. Features a tapered connection for the head. Made from titanium or cobalt-chromium alloys. |
Expertise in orthopedic implants and instruments manufacturing, supplying to over 2,500 clients in 70+ countries for over 15 years.

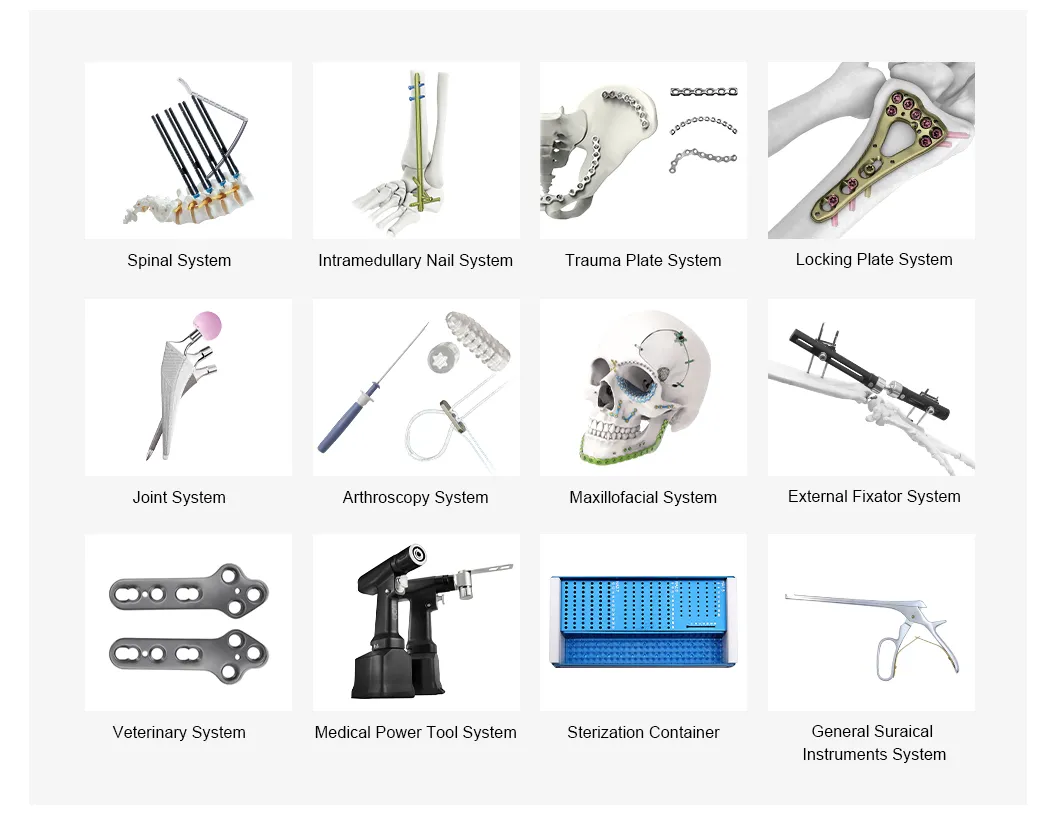
Comprehensive portfolio spanning orthopedics, trauma care, veterinary medicine, and sterilization.
CE and ISO 13485 certified quality management systems.

2025.01 | UAE

2024.11 | Germany

2024.10 | Indonesia
