

1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 119/Piece Request Sample |
| Model NO. | T1100-38 |
| Group | Adult |
| Delivery Time | Within 7 Days |
| Shipping | DHL, UPS, TNT, FedEx, Aramex, EMS |
| MOQ | 1 Piece |
| OEM | Accepted |
| Transport Package | PE Film |
| Origin | China |


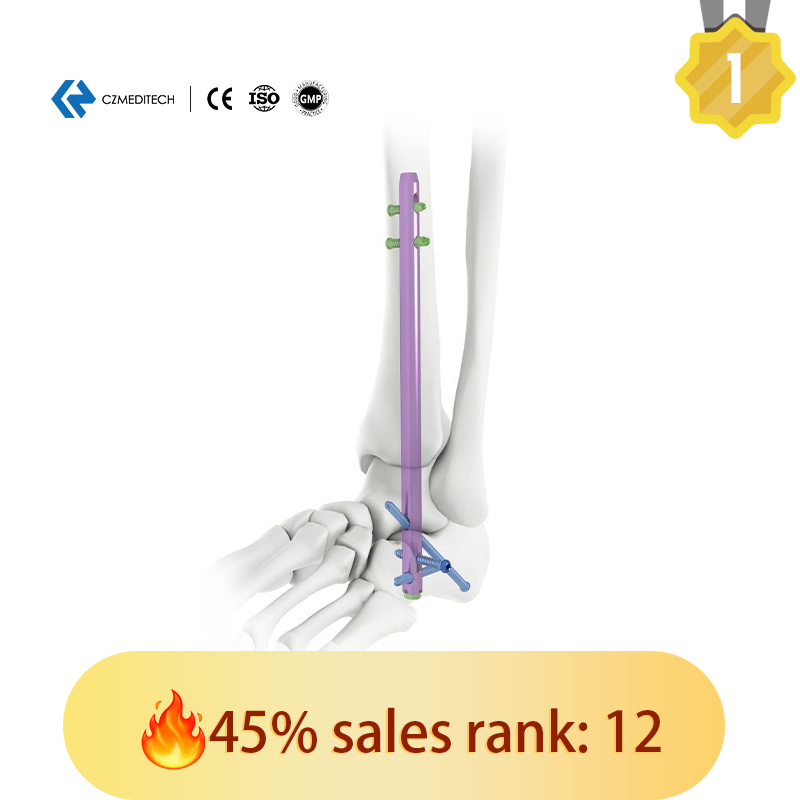
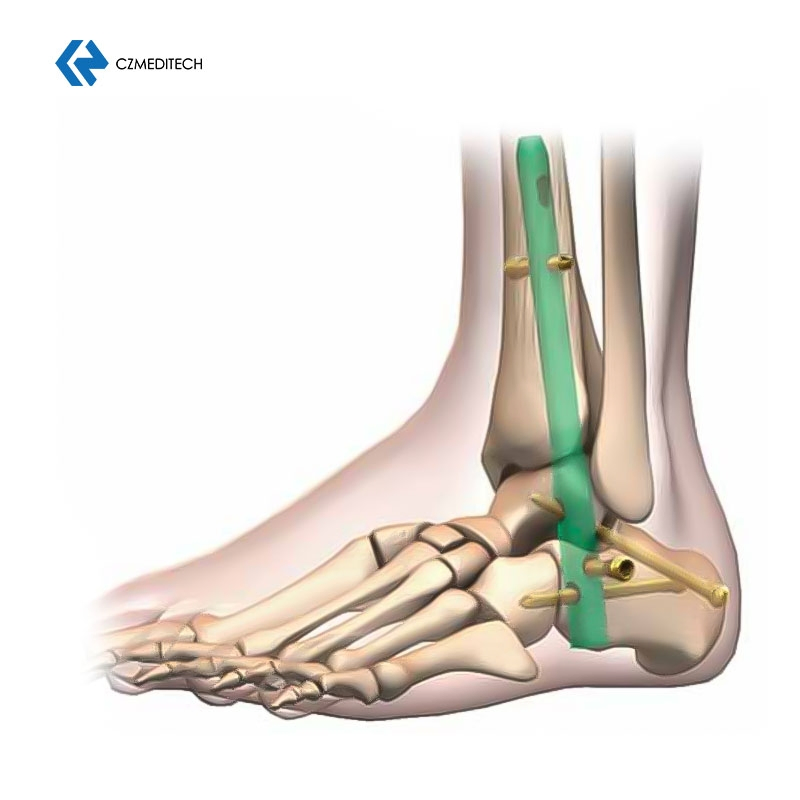
This titanium intramedullary nail is specifically designed for hindfoot and ankle fusion procedures, including tibiotalocalcaneal (TTC) arthrodesis. The interlocking design provides excellent rotational and axial stability, promoting successful fusion.


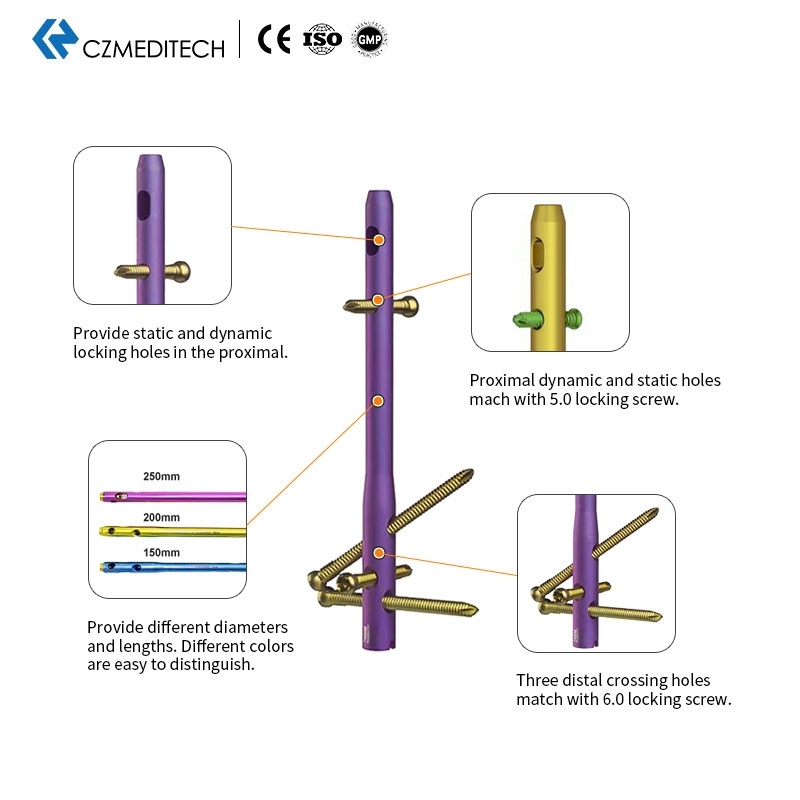
Color-Coded Selection
Different diameters and lengths are color-coded for easy surgical identification.
Locking Versatility
Provides both static and dynamic locking holes in the proximal section.


| Product Item | Available Sizes (Diameter * Length) |
|---|---|
| Hindfoot Fusion Ankle Nail | 10mm * 150/200/225/250 mm (Left & Right) |
| 11mm * 150/200/225/250 mm (Left & Right) | |
| 12mm * 150/200/250 mm (Left & Right) | |
| 5.0mm Locking Screws | Length: 28mm up to 60mm |
| 6.0mm Locking Screws | Length: 26mm up to 100mm |
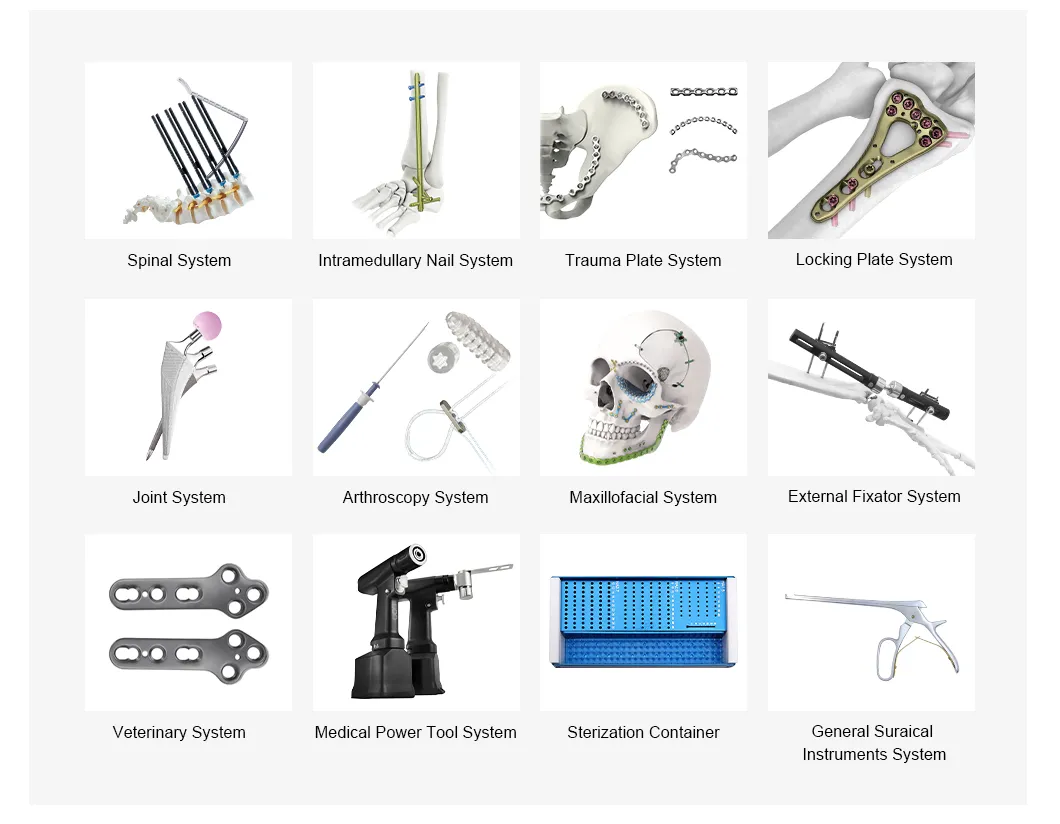
We provide a comprehensive product portfolio spanning orthopedics, trauma care, and sterilization. Our quality management system is ISO 13485 certified, and all products carry CE certification.

ARAB HEALTH (UAE)

MEDICA (Germany)

FIME (USA)