📋 Basic Information
Implant Type
Intramedullary Nail
Specification
Titanium Alloy TC4
🔬 Product Introduction
The PFNA (Proximal Femoral Nail Anti-rotation) is an advanced intramedullary implant specifically designed for the treatment of hip fractures, particularly unstable intertrochanteric and proximal femoral fractures. This system achieves stable fixation by sharing the load with the bone through the intramedullary nail, significantly reducing the risk of implant loosening and failure. Made from titanium alloy, PFNA combines excellent biocompatibility with mechanical strength to ensure long-term durability and patient safety.
🌟 Key Features
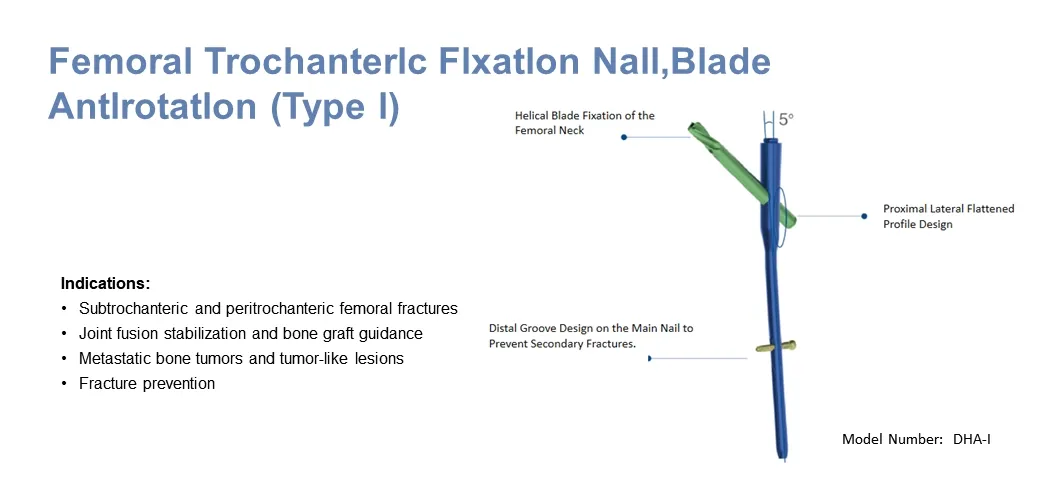
- Helical Blade Design: The unique helical blade compacts cancellous bone, providing rotational and angular stability, especially beneficial for osteoporotic patients.
- Anti-Rotation Mechanism: The blade prevents rotational movement of the nail within the femur, ensuring stable fracture healing.
- Locking System: Distal locking options provide axial stability with dynamic or static locking modes.
- Anatomic Design: 5° outward deviation and 1500mm front arch, perfectly matching the physiological structure of the human body.
- Minimally Invasive: The elastic tip and groove design facilitates the insertion of the main pin and achieves ideal stress distribution.
✅ Clinical Advantages
- Strong Rotational Stability: Effectively prevents rotational displacement of the fracture fragments.
- Load Sharing Design: Decreases implant fatigue and fracture risk.
- Suitable for Osteoporotic Bone: Blade compaction enhances fixation in patients with poor bone quality.
- Early Mobilization: Supports early postoperative weight-bearing, shortening hospital stay.
🏥 Clinical Applications
- Unstable intertrochanteric fractures (AO/OTA 31-A1, 31-A2)
- Subtrochanteric fractures (AO/OTA 31-A3)
- High and low subtrochanteric fractures and femoral shaft fractures
- Pathological fractures and complex multiple fractures
❓ Frequently Asked Questions
What are the shipping methods and associated costs?
We typically utilize courier services for shipping, but we can also accommodate postal services upon your request. Shipping costs are determined by the destination, package size, and weight, and we provide a detailed quote based on specific requirements.
Are the orthopedic products certified with ISO 13485?
Yes, our manufacturing processes hold the EN ISO 13485 certification, ensuring that the products meet stringent international requirements for safety and performance in the medical device industry.
What is the minimum order quantity (MOQ) for international orders?
Our standard minimum order value is US$1000. However, we offer sample orders at cost to allow potential clients to evaluate product quality before committing to larger volumes.
Can you provide custom products not listed in the standard catalogue?
Yes, custom orders are accommodated. By providing pictures or samples of the desired product, we can work to create a similar item. Please note that custom orders may have a longer lead time.
Do you maintain stock for immediate delivery?
We maintain stock for most products with regular monthly production cycles. However, stock levels vary based on market demand. It is best to consult regarding current inventory for specific items.
What material is used for the PFNA implants?
The implants are manufactured from high-grade Titanium Alloy (Ti-6Al-7Nb or TC4), which balances exceptional mechanical strength with excellent biocompatibility.





 Blutree Orthopedic Implants
Blutree Orthopedic Implants