Basic Specifications
Model NO.
Spinal Fixation System
Material
Pure Titanium/Titanium Alloy/Stainless Steel
Product Introduction
Key Features
- Immediate lamina supporting in surgery, best maintain the canal plasty expanded.
- The lower dentate supporting plate provides a more stable supporting force.
- Arc openings provide better inclusion.
- Has certain plasticity, can be slightly moulded in operation.
Main Advantages
- Provide a stable support for vertebral body decompression.
- Four holes design optimized for surgery requirements.
- Unique angle makes the vertebral body more stable.
Product Parameters
| PRODUCT NAME |
PRODUCT CODE |
DIA |
DATA (mm) |
| Laminoplasty Plate |
11101012 |
/ |
H=12 |
| 11101014 |
/ |
H=14 |
| 11101016 |
/ |
H=16 |
| Screw (Cross self-tapping) |
10511306 |
φ2.5 |
L=6 |
| 10511308 |
L=8 |
| 10511310 |
L=10 |
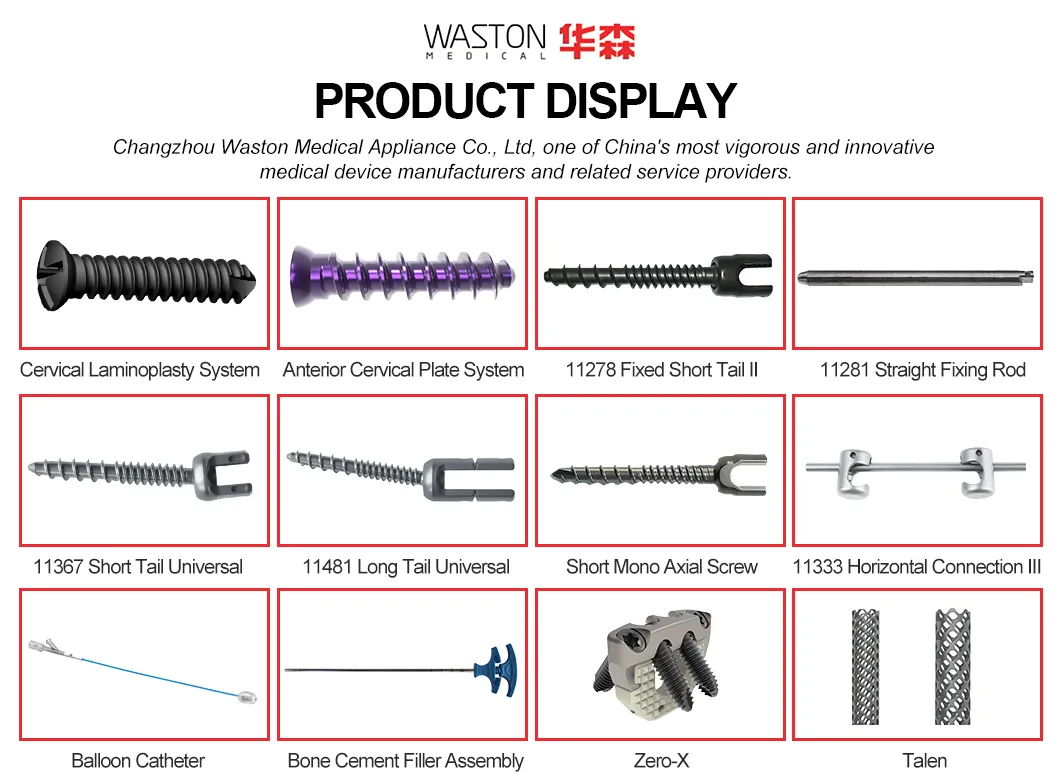
Product Recommendation Display
Manufacturing & Quality Control
As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. We pioneer globally revolutionary innovation technologies such as the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plates, which are fully endorsed by experts and the market.
Our facility operates strictly according to the ISO13485 quality system and medical device manufacturing quality management rules, following CE MDD 93/42/EEC directives. Our Testing Center includes a biomechanical laboratory and a physical-chemical testing center, ensuring control over raw materials, product quality, and performance analysis.
Global Presence & OEM
Our products are exported to many countries, including France, Turkey, Indonesia, Mexico, Russia, and Thailand. We are also a major OEM supplier to some of the largest global players in the orthopaedic industry.
Certifications & Packaging
Frequently Asked Questions
1. Is customized design available for these implants?
Yes, we provide customized design options based on specific requirements and drawings.
2. What is your return policy for defective goods?
For defective goods, please provide photographic evidence for confirmation, and we will arrange for a replacement.
3. Can I request samples to test quality before a bulk order?
Yes, we welcome sample orders to test our product quality and service performance in your local market.
4. What is the typical delivery time for orders?
Samples are generally delivered within 7 days, while standard bulk orders are processed within 25 days.
5. What international certifications do your products hold?
Our products are certified with CE and ISO. The Spinal System also holds FDA registration.
6. Do you maintain inventory for regular orthopaedic items?
We generally keep stock for listed regular items. However, inventory levels fluctuate, so please check with our team for real-time availability.



 Blutree Orthopedic Implants
Blutree Orthopedic Implants