




1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
| Model NO. | Cage System |
|---|---|
| Material | Pure Titanium / Titanium Alloy / Stainless Steel |
| Feature | Disposable |
| Certification | CE, ISO |
| HS Code | 9021100000 |
| Package | Non-Sterile Package, Paper Plastic Roll Bag |

| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 | ||
| NEULEN Screw (Self-drilling) | 10511406 | φ2.5 | L=6 |
| 10511408 | L=8 | ||
| 10511410 | L=10 |
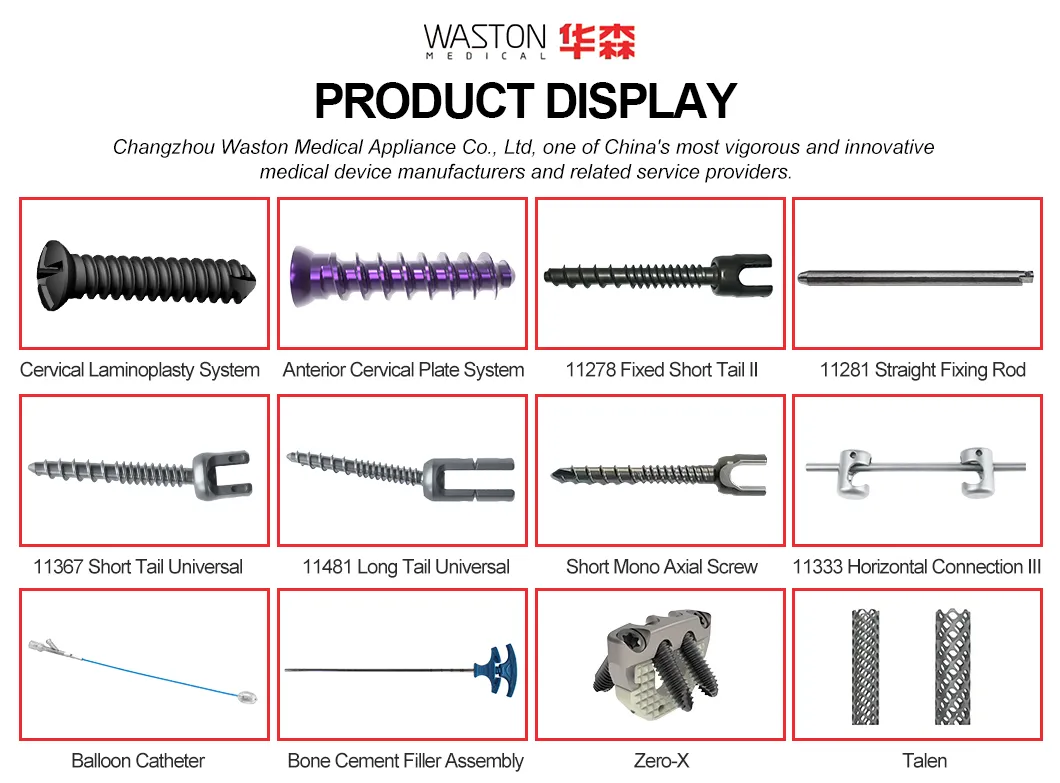

As a national high-tech enterprise, we invest heavily in R&D to transform clinical experience into innovative medical achievements. Our revolutionary systems like the Rev Drill system, Three-Row-Stapler, and Sternal Fixation are endorsed globally by experts and the market.



We operate strictly according to the ISO13485 quality system and CE MDD 93/42/EEC directives. Our testing center includes biomechanical and physical-chemical laboratories to ensure raw material procurement control and product performance analysis.
Our products are exported to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for global leaders in the orthopaedic industry.

