
1 / 5
 Blutree Orthopedic Implants
Blutree Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 30/Piece Request Sample |

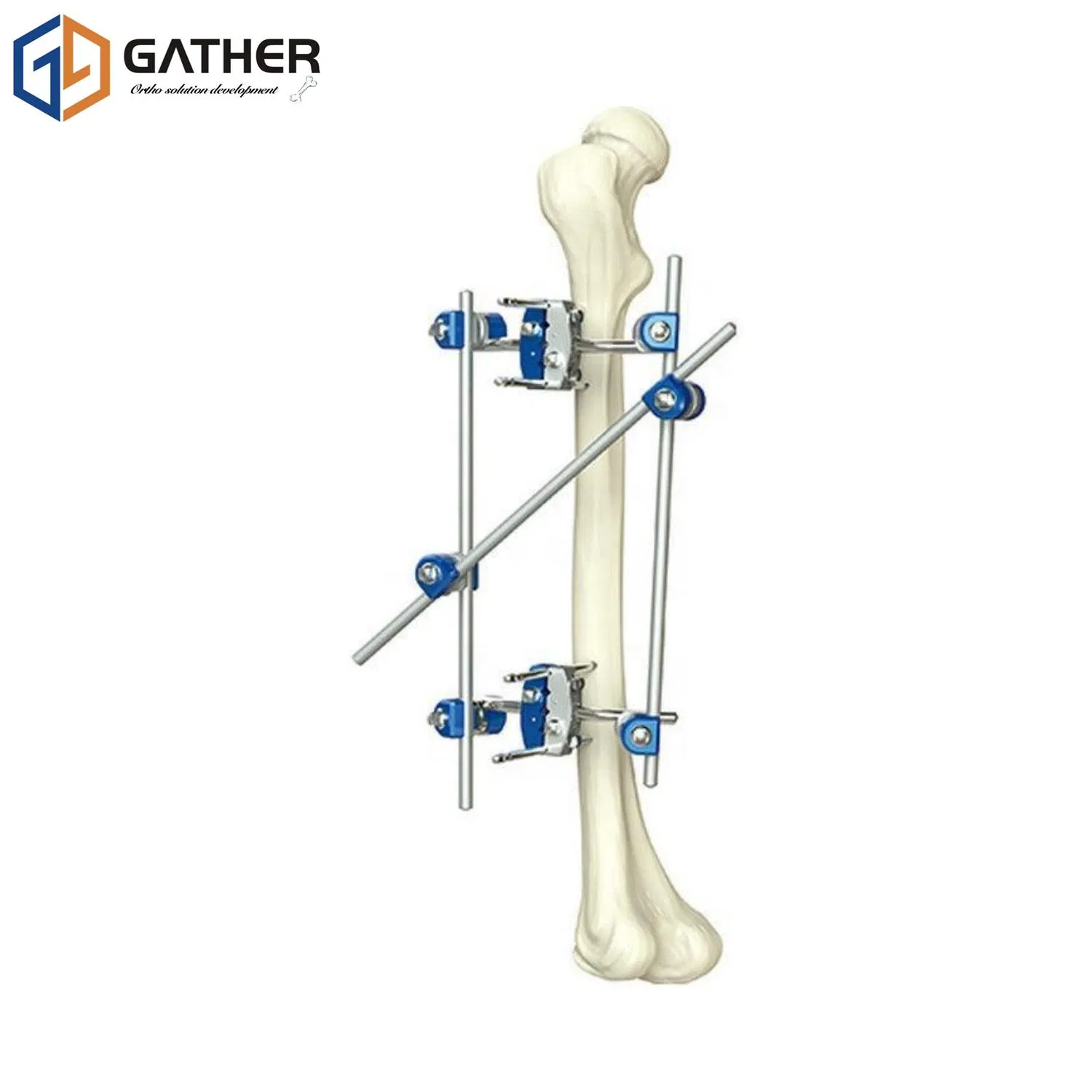
| Name | Orthopedic External Fixator Rod rod climp Upper & Lower Limbs Instruments |
| Material | S.S |
| Valid Time | 3 Years |
| Licence number | CE/ISO9001 |
| Usage | Medical Fixation Equipment |