Basic Overview
Model NO.:
CanETN
Usage:
Tibial Surgery
Transport:
DHL/Express
Origin:
China
HS Code:
9021100000
Tibial Nail Intramedullary Interlocking Cannulated System
Technical Specifications
| Description |
SPEC 1 |
SPEC 2 |
Direction |
| Nail |
ф9-ф12 |
180-240 |
/ |
| Nail |
ф9-ф12 |
180-240 |
Left |
| Nail |
ф9-ф12 |
180-240 |
Right |
| Nail |
Ф9×300 |
300-420 |
Left |
| Nail |
Ф9×300 |
300-420 |
Right |
| Helical Blade |
70-120 |
/ |
/ |
| Compression Screw (Pin) |
60 |
/ |
/ |
| Lag Screw |
70-120 |
/ |
/ |
| Locking Screw |
Φ4.9×28 |
28-80 |
/ |
| End Cap II |
0-15 |
/ |
/ |
Manufacturing Profile
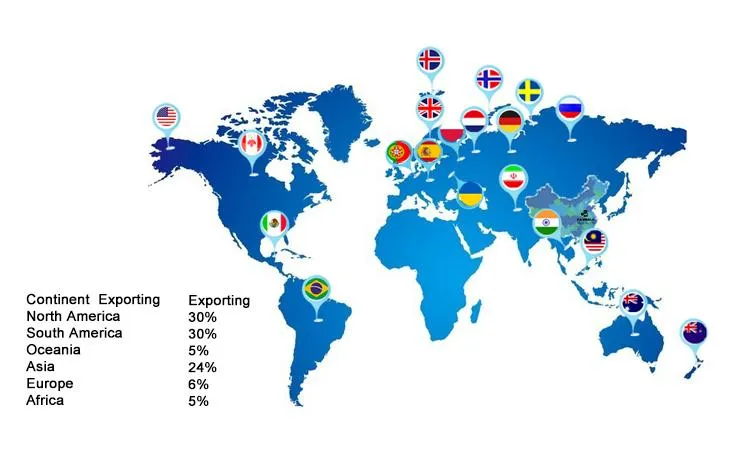
A medical device company founded in 1994 is now one of the largest designers, manufacturers, and suppliers of orthopedic implants and instruments. With over 60,000 square meters of workshop, the company serves as a long-term instrument contract manufacturer for top global orthopedic firms. Product lines include traumatology, spinal fixation systems, and surgical instruments, covering femoral nails, titanium plates, screws, PEEK cages, and balloon kyphoplasty sets, exported to Europe, Latin America, the Middle East, and Asia.
Frequently Asked Questions
What information can you provide regarding shipping modes and charges?
Normally, products are sent via courier or post based on client requests. Shipping charges depend on the weight and destination.
Do you hold international quality certifications such as ISO or CE?
Yes, we are an ISO 9001 and EN ISO 13485 certified company. Many of our products carry the CE mark for compliance with international standards.
What is the minimum order requirement?
While we generally expect minimum orders of at least US$1000, we understand the need for smaller sample orders to test market feasibility. Samples are available upon request.
Can I request products that are similar to your catalog but not specifically listed?
Yes, you can provide pictures or samples. Please note that custom or non-catalog items may require a longer lead time for delivery.
Are the listed products currently in stock?
Generally, we maintain stock for every listed product with monthly production cycles. However, availability depends on sales volume. Please consult our sales team for real-time inventory updates.
How do you ensure the quality of orthopedic implants?
Quality is ensured through rigorous adherence to ISO standards, state-of-the-art manufacturing facilities, and extensive testing protocols throughout the production process.





 Blutree Orthopedic Implants
Blutree Orthopedic Implants