 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System
High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System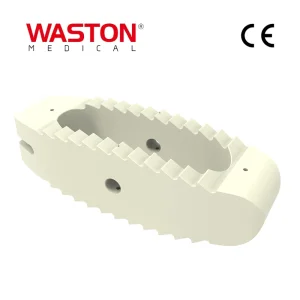 Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System
Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System High Quality Cannulated Axial-Rotation Pedicle Screw Human Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation Minimally Invasive System
High Quality Cannulated Axial-Rotation Pedicle Screw Human Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation Minimally Invasive System Titanium Alloy Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
Titanium Alloy Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System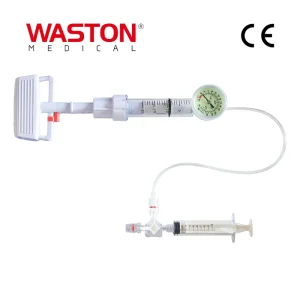 Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Orthopaedic Instrument Implant Apex Balloon Pump Vertebroplasty System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Orthopaedic Instrument Implant Apex Balloon Pump Vertebroplasty SystemModern spinal surgery demands materials that offer the perfect balance between strength and elasticity. While Titanium alloys (Ti-6Al-4V) remain the industry standard due to their excellent biocompatibility and osseointegration properties, the emergence of Cobalt-Chrome (CoCrMo) rods has revolutionized the correction of complex spinal deformities. These high-strength systems provide the stiffness required for massive corrective forces while maintaining a low profile to minimize soft tissue irritation. As a leading supplier, we integrate these material advancements into every screw we manufacture, ensuring that surgeons have the tools they need for the most challenging cases.
Minimally Invasive Surgery (MIS) is the dominant trend. Our cannulated axial-rotation screws are designed for percutaneous insertion, reducing recovery time and blood loss.
Modern pedicle screws are now designed with navigation-ready interfaces, compatible with robotic-arm assisted platforms for sub-millimeter placement accuracy.
Advanced surface treatments, such as hydroxyapatite (HA) coating, are being applied to improve the screw-to-bone interface and prevent long-term loosening.
Procurement needs vary significantly by region. In high-income markets like Western Europe and North America, there is a fierce demand for Single-Use (Disposable) Surgical Kits to reduce sterilization costs and the risk of cross-contamination. Conversely, in rapidly developing regions, durability and versatile instrument sets (like our 6.0mm systems) are prioritized to maximize the utility of hospital resources.
As a factory, we provide localized solutions. For US-based distributors, we offer FDA-aligned documentation and OEM branding. For hospitals in Southeast Asia or Latin America, we provide comprehensive training and complete surgical sets that are compatible with various anatomical profiles.
Integrated supply chains allow us to offer premium titanium implants at a fraction of the cost of Western competitors without compromising on clinical outcomes.
With over 100 global partners, our R&D team can turn a blueprint into a sterile, CE-certified product in record time, tailored to your specific market needs.
Our 214 CE-certified products and ISO 13485 compliance guarantee that every screw passing through our factory meets the highest safety standards.


 Disposable Titanium Alloy Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
Disposable Titanium Alloy Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws
Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Orthopaedic Instrument Implant Wave 61621 Interbody Fusion Cage System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Orthopaedic Instrument Implant Wave 61621 Interbody Fusion Cage System Pedicle Screw System for Spinal Fusion
Pedicle Screw System for Spinal Fusion Pedicle Fusion Plate and Screw System
Pedicle Fusion Plate and Screw System Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Arrow Cage System
Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Arrow Cage System Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant 11412 Fixation Rod IV 6.0 Spinal Fixation System
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant 11412 Fixation Rod IV 6.0 Spinal Fixation System OEM ODM H7 Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant Spinal Fixation System
OEM ODM H7 Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant Spinal Fixation System