 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Precision Engineering & Global E-E-A-T Certified Surgical Solutions for Bissau-Guinean Healthcare Excellence

Guinea-Bissau Specialized Titanium Distal Radius Locking Plate Instruments Set
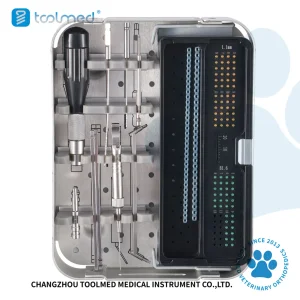
1.6/2.4/3.2/4.0mm ALPS Veterinary Surgical Kit for Bissau Animal Clinics
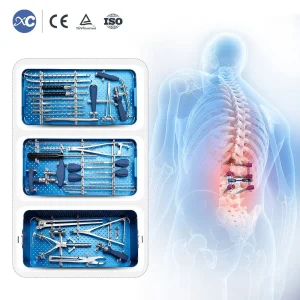
6.0mm Spinal Pedicle Screw System Instrument Set - Guinea-Bissau Trauma Grade

Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm - High-Stability Spine Solution
In the evolving healthcare landscape of Guinea-Bissau, the demand for high-quality orthopedic instrument sets has reached a critical juncture. As the nation invests in upgrading facilities like the Simão Mendes National Hospital and private clinics in Bissau, the need for reliable, CE-certified surgical tools is paramount. Our role as a leading manufacturer is to bridge the gap between global innovation and local clinical needs.
Unlike more developed markets, orthopedic surgery in West Africa often deals with complex trauma resulting from road traffic accidents and delayed presentations. This requires instrument sets that are not only precise but exceptionally durable and easy to maintain under varying sterilization conditions. Our titanium alloy sets are specifically engineered for high-torque resistance and biocompatibility to reduce post-operative complications in resource-constrained environments.
Our instruments are designed to be compatible with both modern digital operating theaters and traditional surgical setups found across Guinea-Bissau's regions.
Using medical-grade Titanium and Stainless Steel (316LVM), we ensure that every drill bit, tap, and screwdriver meets international ISO 13485 standards.
Recognizing the logistical hurdles in Bissau, we provide specialized "Trauma Kits" that include redundant essential components to ensure surgery proceeds even if an individual tool is compromised.
The global orthopedic instrument market is shifting toward minimally invasive surgery (MIS) and smart instrumentation. As a manufacturer with over 25 years of experience, we are at the forefront of this transition, ensuring our partners in Guinea-Bissau have access to "Future-Ready" technology.
1. Surface Engineering: We are implementing advanced anodic oxidation on titanium instruments to improve wear resistance and color-coding for surgical speed.
2. Ergonomic Redesign: Modern sets feature silicone-molded handles that reduce surgeon fatigue during long reconstructive procedures, a vital factor in high-pressure surgical environments.
3. Sustainability in Sterilization: Our instrument trays are designed for optimal steam penetration, reducing sterilization cycles and energy consumption for hospitals.
Founded in 1999, our facility integrates AI-driven CNC machining with rigorous manual checking to ensure zero-defect delivery.
 CNC Machining
CNC Machining Tumbling
Tumbling Polishing
Polishing Washing
Washing Quality Checking
Quality Checking Packing
Packing Wire Cutting
Wire Cutting Slitting Machine
Slitting MachineEvery batch of instrument sets destined for Guinea-Bissau undergoes three-stage validation:
We provide full surgical suites including implants, dedicated instruments, and sterilization containers, reducing the complexity of managing multiple suppliers in Guinea-Bissau.
Offering digital training manuals and video guides for surgical technicians in Bissau to master the locking plate systems and spinal fixation techniques.
With local registration already obtained in over 60 countries, we assist Guinea-Bissau importers with all necessary documentation (CE, ISO, FSC) for rapid customs clearance.

2025 Toolmed ALPS Reconstruction Locking Plate Kit for Bissau Veterinary Excellence

Knee Reconstruction ACL Surgery Arthroscopy Instrument Set for Sports Medicine

Complete Ligament Repair Kit - Acl Pcl Reconstruction Instrument Set

Titanium PFNA Gamma Intramedullary Nail Trauma Set for Femoral Fractures

6.5mm Cannulated Screw Instrument Set - High-Precision Orthopedic Fixation

5.5 Pedicle Screw Lumbar Posterior Titanium Rod Installation Set

Posterior Cervical Surgery Orthopedic Spine Surgical Instruments Set

Titanium Maxillofacial Orthopedic Plate Surgical Instrument Set
All our products are manufactured under ISO 13485 quality management systems and have obtained CE certification. They meet and often exceed ASTM and ISO metallurgical standards for medical devices.
Yes, we have a robust global logistics network. We handle air and sea freight to Bissau, providing all necessary documentation for West African customs clearance.
While optimized for our own implants, many of our general trauma and spinal instruments are designed with universal sizing to be compatible with standard AO-style implants.
Absolutely. We partner with over 100 global OEM clients, providing customized branding and packaging solutions for local distributors in Guinea-Bissau who wish to build their own medical brand.