 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Leading the way in traumatology with localized precision and international quality standards.
 Ho Chi Minh City Special: Gustilo Revdrill and Controller, Femoral Intramedullary Nail
Ho Chi Minh City Special: Gustilo Revdrill and Controller, Femoral Intramedullary Nail
 Surgical Implant Tibia Femural Fracture Fixation Device Orthopedic Interlocking Intramedullary Nails
Surgical Implant Tibia Femural Fracture Fixation Device Orthopedic Interlocking Intramedullary Nails
 Titanium Orthopedic Femur Interlocking Nail Medical Bone Surgical Femoral Intramedullary Implant
Titanium Orthopedic Femur Interlocking Nail Medical Bone Surgical Femoral Intramedullary Implant
 High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
Ho Chi Minh City (HCMC) has emerged as the beating heart of Vietnam's medical device industry. As the nation's largest economic hub, the city provides a sophisticated infrastructure that supports high-precision manufacturing, particularly in the specialized field of Intramedullary (IM) Nails. The industrial parks surrounding the city, such as the Saigon Hi-Tech Park (SHTP), have become focal points for biomedical engineering, attracting both domestic innovators and international manufacturers.
The demand for intramedullary nails in Ho Chi Minh City is driven by two critical factors: a rising middle class seeking better healthcare outcomes and the city's role as a regional trauma center for Southern Vietnam. Hospitals like Cho Ray Hospital and the HCMC Hospital for Traumatology and Orthopedics perform thousands of fixations annually, necessitating a robust supply chain of high-quality femoral, tibial, and humeral interlocking nails. This local demand has catalyzed a shift from mere distribution to advanced manufacturing and R&D within the city limits.
Manufacturers in Ho Chi Minh City are increasingly adopting Industry 4.0 technologies. From CNC precision milling to automated surface treatment processes like anodization (which creates the distinct blue, gold, and green colors seen in titanium implants), the local factory standards are aligning with global expectations. This evolution ensures that orthopedic surgeons in Vietnam have access to tools that are not only biocompatible but also designed with the specific anatomical nuances of the Asian population in mind.
Founded in 1999, we are pioneers in orthopedic implant research and development.
With more than 3 overseas offices established in the United States and the Netherlands, our influence extends far beyond Ho Chi Minh City. We bridge the gap between world-class engineering and local healthcare needs.
The field of traumatology is undergoing a rapid digital transformation. In Ho Chi Minh City, manufacturers are exploring the integration of AI-assisted design for intramedullary nails. By analyzing vast datasets of bone morphology, engineers can optimize the curvature and locking hole placement of nails to ensure a better "fit and fill" within the medullary canal. This is particularly vital for the Proximal Femoral Titanium Nail (PFNA), where precision in the trochanteric region is paramount for elderly patients with osteoporotic fractures.
Another significant trend is the move toward "Smart Implants." While still in the developmental phase in many HCMC labs, the concept involves sensors within the interlocking nail that can monitor bone healing and weight-bearing progress. Furthermore, the use of Medical-Grade Titanium Alloys (Ti6Al4V) has become the gold standard in Ho Chi Minh City factories, offering superior strength-to-weight ratios and excellent fatigue resistance compared to traditional stainless steel.
Surface technology is also evolving. Modern intramedullary nails now feature specialized micro-coatings that promote faster osseointegration and reduce the risk of implant-associated infections—a critical concern in tropical climates. These innovations are positioning HCMC-based suppliers as competitive players in the broader Southeast Asian medical market.
In the bustling streets of Ho Chi Minh City, traffic-related trauma remains a significant public health challenge. High-energy fractures of the femur and tibia are common, requiring immediate surgical intervention with interlocking intramedullary nails. Our products, such as the Multi-Screw Hole Tibial Nail, are specifically designed to handle these complex multi-fragmentary fractures, providing the rotational stability needed for early mobilization.
Furthermore, Vietnam is experiencing an aging population demographic shift. This has led to an increase in hip fractures among the elderly in HCMC. The Gama Interlocking PFNA Nail has become a staple in local geriatric orthopedic care, allowing for minimally invasive procedures that reduce blood loss and hospital stay durations. By manufacturing these nails locally or through dedicated OEM partnerships, we ensure that hospitals in District 1, District 10, and Thu Duc City have constant access to life-saving implants.


Discover our full range of surgical implants designed for the HCMC medical market.
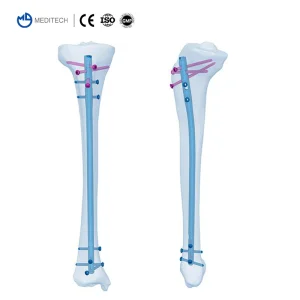 Direct Sale Orthopedic Implant Titanium Expert Tibia Interlocking Intramedullary Nail
Direct Sale Orthopedic Implant Titanium Expert Tibia Interlocking Intramedullary Nail
 HCMC Infrastructure Choice: Multi-Screw Hole Tibial Intramedullary Nail
HCMC Infrastructure Choice: Multi-Screw Hole Tibial Intramedullary Nail
 Factory Instrument Expert Femur Intramedullary Nail Orthopedic Intramedullary Nails for Human
Factory Instrument Expert Femur Intramedullary Nail Orthopedic Intramedullary Nails for Human
 Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200
Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200
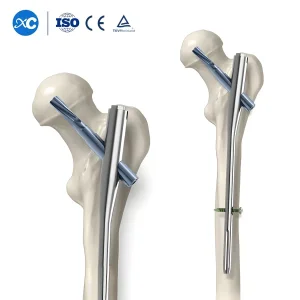 Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
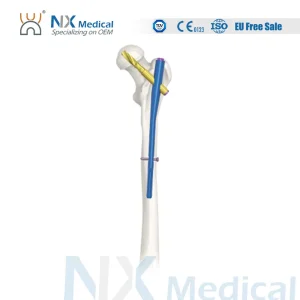 Nx Medical Orthopedic Implant Pfna Proximal Femoral Titanium Intramedullary Nail
Nx Medical Orthopedic Implant Pfna Proximal Femoral Titanium Intramedullary Nail
 Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail
Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail
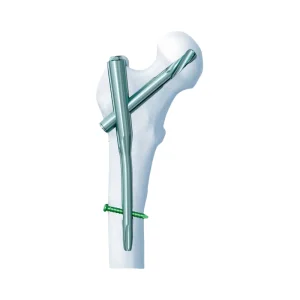 Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral
Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral
The strategic location of Ho Chi Minh City makes it an ideal export hub for intramedullary nails targeting Southeast Asian, European, and American markets. With the signing of several free trade agreements (FTAs), Vietnam's medical manufacturing sector is more connected than ever. Companies that manufacture intramedullary nails in Ho Chi Minh City benefit from a skilled workforce, lower operational costs, and proximity to major shipping ports like Cat Lai.
Looking ahead to 2030, the city's medical landscape will likely be defined by "precision and personalization." Custom 3D-printed intramedullary nails, while currently a niche, are on the horizon. These implants will be designed from CT scans of the individual patient, providing a perfect anatomical fit that reduces operative time and improves long-term clinical outcomes. Our factory is at the forefront of this evolution, investing in the machinery and training necessary to bring these advanced technologies to the surgeons and patients of Ho Chi Minh City.
Whether you are a hospital purchasing manager, a distributor looking for high-quality Humeral Interlocking Nails, or a global brand seeking an OEM partner, the medical cluster in Ho Chi Minh City offers unparalleled value. Our commitment to CE certification and ISO standards ensures that every intramedullary nail we produce is a masterpiece of biomedical engineering.