 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
The global market for Intramedullary Nails (IMN) has witnessed unprecedented growth over the last decade. As the gold standard for treating long bone fractures—specifically in the femur, tibia, and humerus—intramedullary nailing offers superior biomechanical advantages over traditional plating. By acting as a load-sharing device, these nails allow for earlier weight-bearing and accelerated rehabilitation for patients worldwide.
Modern Intramedullary Nails Manufacturers are shifting from standard stainless steel to high-grade Titanium alloys (Ti6Al4V ELI). These materials offer better biocompatibility and a lower modulus of elasticity, which reduces the "stress shielding" effect. Factories are now integrating advanced surface treatments such as anodic oxidation to enhance fatigue strength and color-code different diameters for surgical convenience.
In the current industrial scenario, the demand is driven by an aging global population prone to hip fractures and a rise in road traffic accidents in developing economies. This has forced factories to scale production while maintaining rigorous ISO 13485 quality standards.
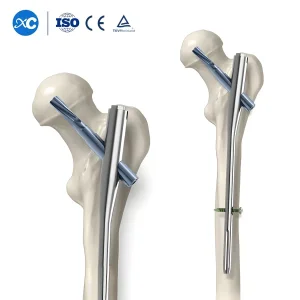
Click to view: Trauma Surgery Implants Gama Interlocking Pfna Nail
New designs focus on reducing the incision size and muscle trauma. Expert nail systems with specialized insertion handles allow surgeons to perform precise interlocking with minimal fluoroscopy exposure.
While Titanium is king, research into radiolucent carbon-fiber-reinforced PEEK nails is gaining traction, allowing for better post-operative X-ray visualization of the callus formation.
The integration of AI and sensor technology into orthopedic implants is on the horizon. Future nails may monitor healing progress and strain distribution in real-time, alerting doctors to potential non-union.
Hospitals and medical distributors in North America, Europe, and Southeast Asia have distinct procurement needs. While developed markets prioritize FDA/CE certified high-end systems, emerging markets look for a balance between cost-efficiency and clinical reliability.
China has emerged as a global hub for medical device manufacturing. Our facility, established in 1999, exemplifies the strengths of Chinese orthopedic production:
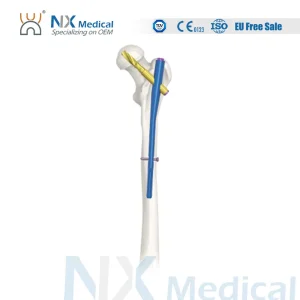
Click to view: Nx Medical Proximal Femoral Titanium Nail
Intramedullary nailing is not a one-size-fits-all solution. Different clinical environments require specific instrument sets and nail variations:
Use of PFNA (Proximal Femoral Nail Antirotation) for osteoporotic hip fractures in elderly patients to ensure stable fixation and prevent cut-out.
Titanium tibial nails for high-impact sports fractures, allowing athletes to begin muscle rehabilitation sooner.
Flexible elastic nails for children's long bone fractures that respect the growth plates (epiphysis).
Simple, robust interlocking systems designed for rapid deployment in military or disaster relief scenarios.
Our manufacturing process involves Swiss-made CNC machining centers that ensure tolerances within microns. This is critical for the "Expert" series of nails where multi-planar locking holes are utilized to secure complex comminuted fractures. The cannulated design allows for reamed insertion over a guide wire, which is preferred for maximizing the contact area between the nail and the endosteum.
From the Proximal Femoral Nail Antirotation (PFNA) to the Retrograde Femoral Nail, each product undergoes rigorous biomechanical testing, including static and dynamic fatigue tests, ensuring they can withstand the repetitive stresses of human gait during the healing phase.
Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas.
Sales channels cover more than 160 countries and regions
160+
Local registration has been obtained in more than 60 countries and regions
60+
More than 3 overseas offices have been established in countries such as the United States and the Netherlands
3+
More than 100 global OEM partners
100+
Currently, 214 products have obtained CE certification
214
