 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
🌐 The global medical community has witnessed a paradigm shift in spinal care. Percutaneous Vertebroplasty (PVP) and Kyphoplasty (PKP) have become the gold standards for treating Osteoporotic Vertebral Compression Fractures (OVCFs). As the world's population ages, the demand for high-precision, reliable Vertebroplasty Kits has surged. As a leading Vertebroplasty Kit manufacturer, we understand that these instruments are not just tools; they are life-changing devices that restore mobility and alleviate chronic pain for millions of elderly patients worldwide.
The industrial status of Vertebroplasty has moved from experimental trials in the late 1980s to a highly sophisticated, multi-billion dollar sector. Modern kits now integrate biocompatible materials, ergonomic delivery systems, and real-time imaging compatibility. This technological maturity has allowed clinics from New York to Shanghai to perform these procedures with outpatient efficiency.
The next generation of Vertebroplasty Kits is focusing on "smart" bone cement (PMMA). These materials are being engineered to release osteogenic factors or antibiotics, transforming the cement from a structural filler into a biological healing agent. Our R&D team is actively exploring these integrations to stay ahead of global clinical needs.
Minimal invasiveness is reaching new heights through robotics. Modern kits are being designed to interface seamlessly with robotic arms, ensuring sub-millimeter precision in needle placement and cement distribution, significantly reducing the risk of cement leakage—a primary concern for surgeons globally.
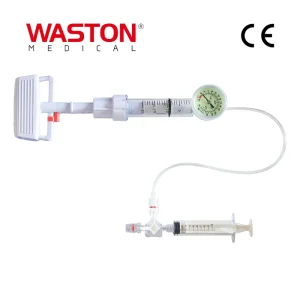
Our flagship Apex Balloon Pump Vertebroplasty System represents the pinnacle of pressure-controlled vertebral restoration. Designed for maximum safety and ease of use, this system allows surgeons to create a void in the collapsed vertebra before injecting high-viscosity bone cement. The kit includes precision-engineered dilators and access needles that minimize soft tissue trauma.
For more details on this system, visit: View Product Specifications
📦 Global procurement officers for hospitals and medical distribution groups look for three critical factors: Regulatory Compliance, Supply Chain Stability, and Cost-Effectiveness. As a specialized China factory, we have aligned our production lines with international standards including CE and ISO 13485.
In Europe and North America, the demand is heavily skewed toward Single-Use (Disposable) Kits to eliminate cross-contamination risks and streamline sterilization logistics. Meanwhile, in emerging markets, there is a strong focus on the durability and versatility of the orthopedic instruments, requiring kits that can perform across various patient anatomies. Our factory's ability to provide 100+ Global OEM solutions allows us to tailor kits specifically to regional medical preferences.
🏥 The application of Vertebroplasty Kits varies significantly by environment. In high-tech surgical suites, our kits are used alongside 3D C-arms and navigation systems. The low-profile needle design of our Vertebroplasty System is ideal for these scenarios as it provides clear visibility under fluoroscopy.
Conversely, in veterinary applications—a rapidly growing niche—the ALPS (Advanced Locking Plate System) is increasingly used for spinal stabilization in large animals. This crossover between human and veterinary surgical needs highlights the versatility of our manufacturing capabilities. Whether it is a routine geriatric procedure in a city hospital or a complex reconstruction in a specialized veterinary center, our tools are engineered for reliability.

When stability is the primary concern, our Titanium Coating Surgical Instrument Sets offer unparalleled durability and biocompatibility. These kits are often paired with Vertebroplasty systems when multi-level spinal fusion is required. The PEEK cage system integrated within this kit ensures optimal bone ingrowth and long-term structural integrity.
Explore our spine implant technology: Detailed Product Link
Since 1999, we have leveraged China's industrial infrastructure to accelerate the R&D cycle. We can transition from clinician feedback to prototype testing faster than many Western counterparts.
By optimizing the manufacturing process and localizing material sourcing, we offer high-grade medical devices at a price point that makes advanced spine surgery accessible to broader patient demographics.
With offices in the USA and Netherlands, we bridge the gap between Chinese manufacturing excellence and local regulatory/logistical requirements.
Founded in 1999, our company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants. We have evolved into a global leader, with products exported to every corner of the world.

