 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Engineered for excellence: Browse our top-selling trauma instrument kits designed for high-stakes surgical environments.
 Medical Instrument Hip Joint Instrument Set Stem Surgical Instrument Acetabular
Medical Instrument Hip Joint Instrument Set Stem Surgical Instrument Acetabular
 Orthopedic Trauma Endoscope Instrument Set for Bone Surgery Surgical OEM Stainless Steel
Orthopedic Trauma Endoscope Instrument Set for Bone Surgery Surgical OEM Stainless Steel
 Orthopedic Mini Instrument for Hand and Foot Surgery Instrument Set Surgical
Orthopedic Mini Instrument for Hand and Foot Surgery Instrument Set Surgical
 High Quality Medical Orthopedic Trauma Titanium Implants Distal Tibiofibular Instrument Set
High Quality Medical Orthopedic Trauma Titanium Implants Distal Tibiofibular Instrument Set
 High Quality Medical Orthopedic Trauma Titanium Implants Distal Tibiofibular Instrument Set
High Quality Medical Orthopedic Trauma Titanium Implants Distal Tibiofibular Instrument Set
 Surgical Instrument Sets Hip Joint Instrument Set for Othopedic Surgery Medical
Surgical Instrument Sets Hip Joint Instrument Set for Othopedic Surgery Medical
 Hand and Foot Instrument Set, Orthopedic Set, Surgical Products
Hand and Foot Instrument Set, Orthopedic Set, Surgical Products
 Medical Orthopedic C Clamp Pelvic Instrument Set Surgical Instrument
Medical Orthopedic C Clamp Pelvic Instrument Set Surgical Instrument
As a leading China trauma instrument kit exporter, we have established a footprint that spans from Europe to Southeast Asia. Our infrastructure includes 3+ overseas offices in the United States and the Netherlands to provide localized support.
Since 1999, our R&D focus has been on high-grade stainless steel and titanium alloys. Every trauma instrument kit undergoes rigorous testing to meet international biomedical standards.
Our facility is one of China's earliest orthopedic implant enterprises. We combine legacy expertise with modern AI-driven manufacturing to produce over 200+ CE-certified items.
The global Trauma Instrument Kit market is currently experiencing a transformative phase. With the rise in geriatric populations and an unfortunate increase in road traffic accidents worldwide, the demand for precision-engineered orthopedic tools has reached an all-time high. Surgeons today require more than just basic tools; they demand modularity, ergonomic design, and material durability that can withstand the rigors of complex bone reconstruction.
Modern trauma kits are evolving towards minimally invasive surgery (MIS). Our kits, such as the Orthopedic Trauma Endoscope Instrument Set, reflect this trend by integrating visualization technologies with traditional mechanical tools. The shift from generic sets to anatomical-specific kits (like our Distal Tibiofibular or Femoral Nail sets) allows for faster surgical turnaround times and better patient outcomes. Furthermore, the integration of 3D printing in orthopedic pre-planning has made the compatibility of instrument kits with custom implants a top priority for global procurement departments.
For years, the medical device sector was dominated by Western brands. However, Chinese manufacturers, led by pioneers like our company founded in 1999, have bridged the quality gap through massive R&D investment. Purchasing from a top China trauma instrument kit supplier offers several strategic advantages:
Our trauma kits are designed with versatile "Application Scenarios" in mind:
Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas. We specialize in everything from Hemodialysis and Critical Care to Percutaneous Coronary Intervention (PCI). Our commitment to quality is underscored by our 214 CE-certified products and our presence in more than 60 countries where we hold local medical registrations.

Strategic Partners: We work with industry giants to ensure our instrument kits are compatible with the latest implant technologies worldwide.
Advanced surgical sets for specialized orthopedic procedures, from spinal fixation to ligament reconstruction.
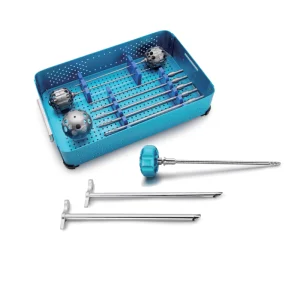 Surgical Endoscope Instrument Set Medical Instrument Set for Orthopedic Surgery
Surgical Endoscope Instrument Set Medical Instrument Set for Orthopedic Surgery
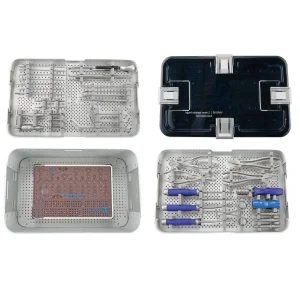 2.4mm Variable Distal Angles Radius Orthopedic Surgical Instrument Sets
2.4mm Variable Distal Angles Radius Orthopedic Surgical Instrument Sets
 Orthopedic Instruments Femoral Nail Instrument Set
Orthopedic Instruments Femoral Nail Instrument Set
 PPSU Al All Kind of Instrument Box
PPSU Al All Kind of Instrument Box
 Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set
 Orthopedic Surgery Trauma Manufacturer Finger External Fixator for Mini Fixator
Orthopedic Surgery Trauma Manufacturer Finger External Fixator for Mini Fixator
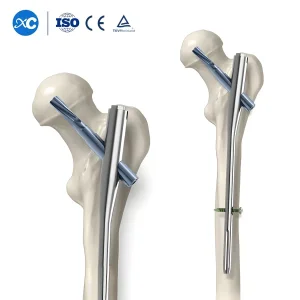 Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
 Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit
Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit
Our company specializes in a wide array of medical disciplines beyond just trauma kits. We provide solutions for Hemodialysis, Critical Care, and Percutaneous Coronary Intervention (PCI). This cross-disciplinary expertise allows us to apply high-tech solutions from cardiovascular science to orthopedic metallurgy.

As one of the earliest enterprises in China to dive into the research and development of orthopedic implants, we have witnessed the evolution of the industry firsthand. Today, we are not just a supplier; we are a strategic manufacturing partner. Our facilities are equipped with CNC machining centers that ensure the precision of every drill, tap, and screwdriver in our trauma kits.
Global buyers can rely on our established sales channels that cover over 160 countries. Whether you are looking for a standard Hip Joint Instrument Set or a complex Variable Angle Radius Set, our portfolio of 214+ CE-marked products ensures you meet the highest clinical requirements.
Q: Are the kits compatible with other implant brands?
A: Yes, many of our Trauma Instrument Kits are designed for universal compatibility or can be customized via our OEM service to match specific implant geometries.
Q: What materials are used in the instruments?
A: We use medical-grade stainless steel (SUS630, SUS440) and aircraft-grade aluminum for the containers, ensuring they can withstand thousands of autoclave cycles.
Q: How do you support local registration?
A: We have a dedicated regulatory affairs team that has successfully registered products in over 60 countries, providing full technical documentation and clinical data support.
Q: What is the average lead time for export?
A: For standard kits, we maintain a rolling inventory for immediate dispatch. Custom OEM orders typically range from 4 to 8 weeks depending on complexity.