 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Heavy-Duty Electroplating Production Line: Custom Gantry Systems
Heavy-Duty Electroplating Production Line: Custom Gantry Systems
 Headless Compression Hollow Screw Cannulated Screw Bone Screws
Headless Compression Hollow Screw Cannulated Screw Bone Screws
 Suture Anchor with Wire Anchor Arthroscopy Titanium & PEEK
Suture Anchor with Wire Anchor Arthroscopy Titanium & PEEK
 Canwell Surgical Cannulated Orthopedic Screw of Trauma Products
Canwell Surgical Cannulated Orthopedic Screw of Trauma Products
 Orthopedic Outer Pillar Brace Osteotomy Locking Plate
Orthopedic Outer Pillar Brace Osteotomy Locking Plate
 Multi-Ring Combined Fixator, Cobra External Fixator
Multi-Ring Combined Fixator, Cobra External Fixator
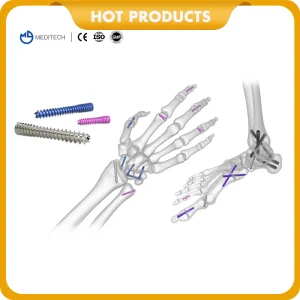
 Orthopedic Implants Cannulated Bone Screws with CE&ISO
Orthopedic Implants Cannulated Bone Screws with CE&ISO
 Cannulated Bone Screws with CE & ISO
Cannulated Bone Screws with CE & ISO
In the rapidly evolving world of orthopedic surgery, Kirschner Wires (K-wires) remain an indispensable tool for surgeons worldwide. These sterilized, sharpened stainless steel or titanium pins are the backbone of fracture fixation, skeletal traction, and temporary bone alignment. As a primary China Kirschner Wire factory, we have observed a massive shift in the global commercial status of these components.
The global Kirschner Wire market is projected to grow at a CAGR of 4.5% over the next decade. This growth is fueled by an aging global population prone to osteoporosis and the increasing prevalence of sports injuries. From North American trauma centers to European veterinary clinics, the demand for high-quality, biocompatible fixation pins is at an all-time high. Modern surgical techniques demand not just a wire, but a precision-engineered instrument capable of withstanding significant mechanical stress while remaining inert within the human body.
Designed for varied load-bearing applications in orthopedic trauma and reconstructive surgery.
Utilizing Medical Grade 316L Stainless Steel and Grade 5 Titanium for maximum biocompatibility.
Fully compliant with CE, ISO 13485, and FDA standards to ensure global patient safety.
The industry is moving toward minimally invasive surgery (MIS). This trend has direct implications for K-wire design. Thinner, stronger wires with specialized tip geometries—such as diamond or trocar points—are being developed to reduce bone fragmentation during insertion. Furthermore, the integration of K-wires with 3D-printed surgical guides is becoming a standard in complex pediatric orthopedic corrections.
International medical distributors and hospital chains are no longer just looking for the lowest price; they are seeking reliability. Global procurement needs have shifted toward manufacturers who can offer high-volume consistency, OEM customization, and localized regulatory support. As a premier Kirschner Wire exporter, we provide end-to-end supply chain transparency, from raw material sourcing to final sterile packaging.
Kirschner Wires find applications across a diverse range of localized scenarios. In human medicine, they are the gold standard for Colles' fracture fixation, phalangeal bone reconstruction, and as guide pins for cannulated screw insertion. Their versatility allows them to be used in virtually every bone in the body, from the smallest joints in the hand to the femoral head.
Interestingly, the veterinary orthopedic sector has seen an explosion in K-wire usage. Small animal practitioners and exotic pet specialists require specialized, miniature fixators and wires for avian wing repairs or reptile limb stabilization. Our Mini External Fixators are specifically designed to work in tandem with our high-precision K-wires to provide stable outcomes for even the smallest patients.


Founded in 1999, our company is one of the earliest enterprises in China dedicated to the research, development, and manufacturing of orthopedic implants. Over the last two decades, we have transformed from a local pioneer into a global powerhouse in the medical device industry.
Our competitive advantage lies in our integration of massive production capacity with R&D agility. Unlike many traders, we control the entire lifecycle of our products. With over 100 global OEM partners and local registrations in 60+ countries, we understand the nuances of different international markets—from the regulatory hurdles in the EU to the logistics challenges in Southeast Asia.
Currently, 214 of our products have obtained CE certification, ensuring that our clients receive instruments that meet the highest safety standards in the world. With offices in the United States and the Netherlands, we provide localized support that bridges the gap between China’s manufacturing excellence and global clinical needs.
 Mini External Fixator for Exotic Pets - Birds & Reptiles
Mini External Fixator for Exotic Pets - Birds & Reptiles
 Veterinary Mini External Fixator for Small Mammals
Veterinary Mini External Fixator for Small Mammals
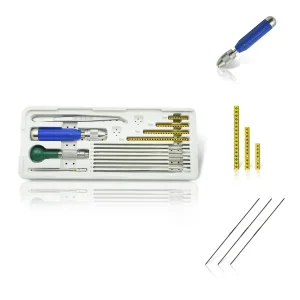
 Vet Clinical Orthopedic Surgical Full Kit K-Wire Pin Set
Vet Clinical Orthopedic Surgical Full Kit K-Wire Pin Set
 Titanium Medical Bone Screws Cannulated Headless Screw
Titanium Medical Bone Screws Cannulated Headless Screw
 Orthopedic Surgical Medical ACL Surgery Instrument Set
Orthopedic Surgical Medical ACL Surgery Instrument Set
 Anterior Posterior Cruciate Ligament ACL/PCL System
Anterior Posterior Cruciate Ligament ACL/PCL System
 Medical Orthopedic C Clamp Pelvic Instrument Set
Medical Orthopedic C Clamp Pelvic Instrument Set
 Custom Gantry Systems for Zinc/Nickel Plating
Custom Gantry Systems for Zinc/Nickel Plating
Choosing the right China Kirschner Wire manufacturer is a decision that impacts surgical outcomes and business profitability. With a robust history dating back to 1999, a global reach across 160+ countries, and a commitment to AI-driven manufacturing precision, we stand as the premier choice for hospital groups and distributors worldwide. Our products, ranging from standard trauma screws to complex external fixation systems, represent the pinnacle of Chinese medical engineering.
Whether you are seeking K-wire exporters for high-volume orders or a strategic OEM partner for innovative orthopedic designs, our team is ready to deliver quality that speaks for itself. Explore our comprehensive product line and join the hundreds of partners who trust us for their surgical essentials.