 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Leading the industry with state-of-the-art trauma and joint reconstruction implants.
The global orthopedic market is witnessing an unprecedented surge in demand for high-performance knee replacement components. At the heart of this revolution is the Tibial Insert, a critical component that acts as the artificial cartilage in Total Knee Arthroplasty (TKA). As the global population ages and the prevalence of osteoarthritis rises, the role of a specialized Tibial Insert Manufacturer has transitioned from a simple supplier to a high-tech engineering partner.
Currently, the market is characterized by a shift towards highly cross-linked polyethylene (HXLPE) and vitamin E-infused materials. These advancements aim to reduce wear rates and prevent osteolysis, extending the life of the implant beyond the traditional 15-20 year window. Hospitals and surgical centers in North America and Europe are increasingly demanding "personalized" tibial inserts that cater to varying degrees of posterior stabilization and cruciate retention.
The global knee reconstruction market is expected to grow at a CAGR of 4.5%, driven by sports injuries and an active elderly demographic.
Transitioning from standard UHMWPE to Antioxidant-Infused Polymers to combat long-term oxidative degradation in the joint.
MDR in Europe and FDA 510(k) in the USA have set new benchmarks for Tibial Insert factories regarding biocompatibility and clinical data.
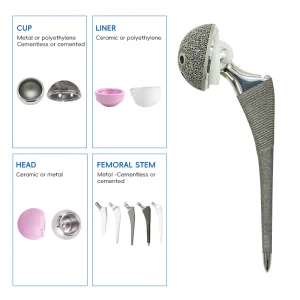
Our premium UHMWPE Tibial Inserts are manufactured using clinical-grade Ultra-High Molecular Weight Polyethylene. These components are designed to provide maximum contact area and minimum peak stress, ensuring smooth articulation and long-term durability. By utilizing advanced CNC machining technology, we achieve tolerances within microns, meeting the exact specifications of modern knee replacement systems.
As a leading Tibial Insert factory, we are closely monitoring several transformative trends that are reshaping the orthopedic industry:
The trend towards minimally invasive surgery (MIS) has also forced manufacturers to develop inserts that can be easily positioned through smaller incisions without compromising the integrity of the locking mechanism between the insert and the tibial tray.
For medical device distributors and hospital procurement departments, sourcing from a Tibial Insert Manufacturer involves more than just comparing prices. The critical factors include:
We address these needs by offering a comprehensive OEM/ODM service, allowing global brands to leverage our manufacturing excellence while maintaining their unique design philosophy.
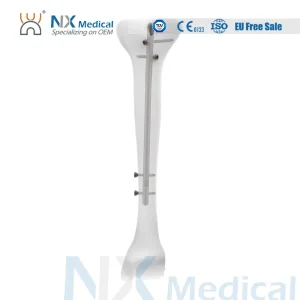
Complementing our soft-tissue inserts, our Tibial Interlocking Nails offer unparalleled stability for tibial shaft fractures. These implants, crafted from high-strength medical titanium alloy, feature a multi-planar locking design to ensure rotational stability and early weight-bearing capabilities for patients.
Founded in 1999, our company has stood the test of time, evolving into one of China's earliest and most sophisticated developers of orthopedic implants. Here is why global partners trust us:
Different regions have unique anatomical requirements. For instance, in many Asian markets, there is a higher demand for High-Flex Tibial Inserts because of cultural practices that involve deep squatting or sitting on the floor. Our factory has developed specific geometries that allow for safe flexion up to 155 degrees without increasing the risk of dislocation or posterior wear.
In Western markets, where BMI (Body Mass Index) can be higher, we focus on reinforced tibial bases and thicker polyethylene inserts to handle higher static and dynamic loads. By offering a modular system, we empower surgeons to choose the best configuration for the specific patient's lifestyle and anatomy.
Corporate Milestone: As a leading enterprise in the research and development of orthopedic implants, we have obtained local registration in more than 60 countries and established overseas offices in the United States and the Netherlands to provide real-time support to our international clientele.

