 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
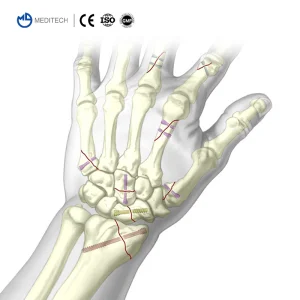
The Herbert screw, a revolutionary headless compression screw designed primarily for fractures of the scaphoid and other small bones, has redefined orthopedic trauma surgery. In today's global medical device market, the Herbert screw industry is witnessing an unprecedented surge in demand. As aging populations in developed nations increase the incidence of bone fractures and the rise of sports medicine in developing regions drives surgical volumes, manufacturers are scaling up precision production. Currently, the global market is dominated by a mix of established Western giants and rapidly advancing Asian manufacturers who offer high-precision, cost-effective alternatives.
From a commercial perspective, the "Top 10 Herbert Screw Manufacturers & Suppliers" are those who have mastered the balance between metallurgical integrity and biomechanical design. Modern Herbert screws are no longer just "fixation devices"; they are sophisticated instruments of healing that utilize variable thread pitches to achieve inter-fragmentary compression without the need for a traditional screw head, which can often cause soft tissue irritation. This unique selling point makes them indispensable in hand, foot, and ankle surgeries globally.
Our flagship Herbert Cannulated Compression Screw is engineered from high-grade Titanium alloy, offering superior biocompatibility and fatigue resistance. Designed for minimally invasive procedures, the cannulated design allows for precise placement over a guide wire, ensuring optimal compression across the fracture site. This product represents the pinnacle of Chinese manufacturing excellence, meeting stringent international standards for orthopedic implants.
Key Advantages: High torque retention, self-tapping tip, and variable pitch threads for controlled compression. Ideal for scaphoid fractures and osteochondral fixations.
The orthopedic implant industry is moving toward "Smart Surgery" and "Bio-Harmonization." Several key trends are shaping the future of Herbert screw manufacturing:
Procurement departments in major hospitals and medical distribution firms have shifted their focus toward "Value-Based Healthcare." They require Herbert screw suppliers who can provide not just a product, but a complete surgical solution including sterilized packaging, precision instrument sets, and comprehensive technical documentation. In the United States and Europe, regulatory compliance (FDA/CE) is the non-negotiable entry barrier, while in emerging markets, the focus is on the price-to-quality ratio.
Localized application scenarios vary: In the EU, there is a high demand for Herbert screws in geriatric care for osteoporotic fractures. In Southeast Asia and Latin America, the demand is driven by high rates of traffic and industrial accidents requiring trauma intervention. As a leading manufacturer, we understand these nuances and offer tailored solutions for every region.
Designed for maximum efficiency, our Headless Compression Hollow Screws allow for a low-profile finish that prevents joint impingement. The hollow core is perfectly matched for guide-wire driven insertion, making it the preferred choice for complex carpal and tarsal fixations. The self-drilling and self-tapping properties reduce surgical steps, minimizing the time the patient is under anesthesia.
Clinical Focus: Perfect for radial head fractures, bunionectomies, and small joint arthrodesis. Available in various lengths and diameters to suit diverse anatomical requirements.

Founded in 1999, our company stands as a pioneer in the Chinese orthopedic implant industry. The "Made in China" label in the medical field has evolved from "low cost" to "high-tech innovation." Our factory integrates the entire value chain—from raw material sourcing of medical-grade titanium to Swiss-style CNC machining and Class 100,000 cleanroom packaging.
Our advantages include:
1. Scalability: With over 100 global OEM partners, we can handle high-volume orders with consistent quality.
2. Regulatory Readiness: 214 of our products have obtained CE certification, and we have local registrations in over 60 countries.
3. R&D Strength: Being one of the earliest enterprises in China, our R&D team works closely with surgeons to iterate designs based on real-world clinical feedback.
4. Global Presence: With offices in the United States and the Netherlands, we bridge the gap between Chinese manufacturing efficiency and Western service standards.

"Dedicated to advancing bone health through precision engineering since 1999."

Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas.
Sales channels cover more than 160 countries and regions
Local registration in 60+ countries
Overseas offices (US, Netherlands)
CE Certified Products