 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
Precision-manufactured implants and instruments for superior clinical outcomes.
 Orthopedic Titanium Alloy Spine Surgical Instrument Set with CE/ISO/FDA
Orthopedic Titanium Alloy Spine Surgical Instrument Set with CE/ISO/FDA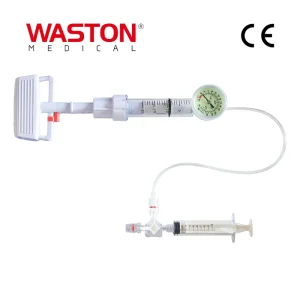 Disposable Surgical Vertebroplasty System / Balloon Pump
Disposable Surgical Vertebroplasty System / Balloon Pump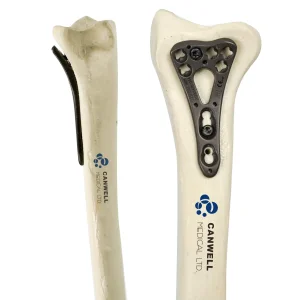 Canwell Variable Angle Radius Locking Plate Orthopaedic Implants
Canwell Variable Angle Radius Locking Plate Orthopaedic Implants Canwell Tibial Nail Intramedullary Nail Interlocking Nail
Canwell Tibial Nail Intramedullary Nail Interlocking Nail Interference Screws for ACL Reconstruction Peek/Cannulated
Interference Screws for ACL Reconstruction Peek/Cannulated Tornillos Canulados, Orthopedic Implants Cannulated Screw System
Tornillos Canulados, Orthopedic Implants Cannulated Screw System Canwell Primary Femoral Bipolar Head Hip Arthroplasty CE
Canwell Primary Femoral Bipolar Head Hip Arthroplasty CE Canwell Orthopedic Trauma Implant Ilizarov Ring External Fixator
Canwell Orthopedic Trauma Implant Ilizarov Ring External FixatorThe global orthopedic market has witnessed a significant paradigm shift toward minimally invasive procedures, with the Kyphoplasty Balloon Catheter standing at the forefront of spinal trauma and geriatric care. As the global population ages, the incidence of Vertebral Compression Fractures (VCFs) caused by osteoporosis has increased exponentially. Kyphoplasty, which involves the inflation of a medical-grade balloon to create a cavity within the vertebral body before injecting bone cement, has become the gold standard for restoring vertebral height and alleviating chronic pain.
Currently, the market is characterized by a high demand for precision and safety. Industrialized nations in Europe and North America are transitioning from traditional vertebroplasty to balloon kyphoplasty due to its ability to reduce cement leakage—a critical complication in spinal surgeries. Emerging markets in the Asia-Pacific region are also seeing rapid adoption, driven by healthcare infrastructure improvements and a growing middle-class population seeking advanced medical treatments.
Innovation in material science, such as the use of high-tenacity polymers and radiopaque markers, allows surgeons to monitor balloon expansion with sub-millimeter accuracy. This technical evolution ensures that Kyphoplasty Balloon Catheters are no longer just tools, but critical components of a digitalized surgical workflow.
Development of next-generation catheters with multi-layered membranes that provide high pressure resistance while maintaining a low crossing profile.
Integrating catheter feedback with real-time AI imaging to predict optimal expansion volume and prevent cortical bone damage.
The trend towards all-in-one surgical kits that include the balloon, access needles, and cement delivery systems for streamlined OR efficiency.
As a leading Kyphoplasty Balloon Catheter supplier in China, we leverage a robust medical device supply chain that offers an unparalleled balance of cost-effectiveness and high-end quality. Founded in 1999, our company has evolved alongside the global orthopedic industry. We don't just manufacture; we innovate. Our R&D centers in China work closely with international orthopedic surgeons to refine the ergonomics and safety of our spinal systems.
Global procurement requirements have become increasingly stringent. Modern buyers—ranging from large hospital groups to specialized distributors—demand full traceability and international certifications. Our facility meets these needs by maintaining CE, ISO, and FDA compliance. We provide comprehensive technical dossiers for local registration, which has allowed us to successfully register our products in over 60 countries and regions.
Our OEM capabilities are a cornerstone of our business. With over 100 global OEM partners, we offer customizable solutions that allow local brands to provide world-class spinal implants without the massive R&D overhead. This collaborative model ensures that patients in every corner of the globe have access to affordable, life-improving spinal technology.
Primary use in elderly patients suffering from painful VCFs where traditional pain management has failed.
Stabilizing the spine in cases of osteolytic lesions caused by multiple myeloma or metastatic disease.
Used in younger patients for specific burst fractures where maintaining vertebral height is crucial for long-term health.
Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas.


Our sales channels cover more than 160 countries and regions. Local registration has been obtained in more than 60 countries and regions, supported by more than 3 overseas offices in countries such as the United States and the Netherlands. With more than 100 global OEM partners and 214 products obtaining CE certification, we are your trusted partner in spinal health.
Extending our expertise beyond the spine into complete orthopedic care.
 Canwell Mini External Fixator Orthopedic Trauma System for Phalanges
Canwell Mini External Fixator Orthopedic Trauma System for Phalanges Orthopedic Shoulder Surgery Arthroscopy Implant Suture Anchors
Orthopedic Shoulder Surgery Arthroscopy Implant Suture Anchors Titanium Sports Medicine Suture Anchor Screw for Arthroscopy
Titanium Sports Medicine Suture Anchor Screw for Arthroscopy Sports Medicine Suture Anchor Shoulder Surgery Rotator Cuff Repair
Sports Medicine Suture Anchor Shoulder Surgery Rotator Cuff Repair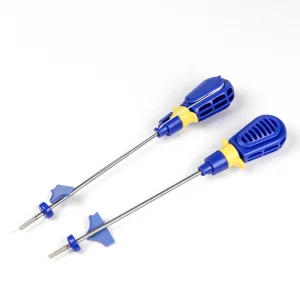 Orthopedic Peek Suture Anchor for Arthroscopic Shoulder Surgery
Orthopedic Peek Suture Anchor for Arthroscopic Shoulder Surgery Canwell Sports Medicine Cartilage Arthroscopy Suture Anchor
Canwell Sports Medicine Cartilage Arthroscopy Suture Anchor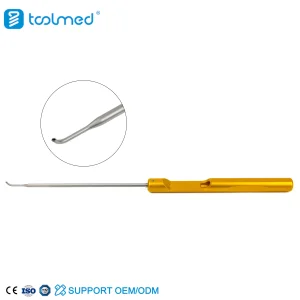 Sports Medicine Arthroscopy System Suture Hook for Surgery
Sports Medicine Arthroscopy System Suture Hook for Surgery Nx Medical Titanium Cannulated Bone Cement Pedicle Screw System
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw System