 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Titanium Pfna Proximal Femoral Intramedullary Nail
Titanium Pfna Proximal Femoral Intramedullary Nail
 Spinal Fixation System Pedicle Screw Instruments Set
Spinal Fixation System Pedicle Screw Instruments Set
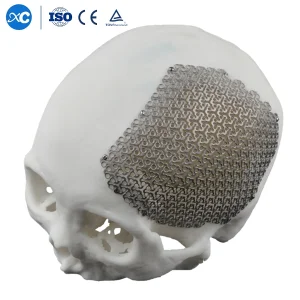 3D Anatomical Cloud Shape Cmf Cranial Mesh
3D Anatomical Cloud Shape Cmf Cranial Mesh
 Mini External Fixator for Exotic Pets
Mini External Fixator for Exotic Pets
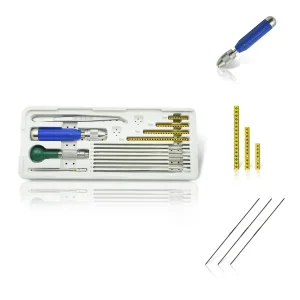 Veterinary Mini External Fixator Fixator
Veterinary Mini External Fixator Fixator
 Trauma Manufacturer Finger External Fixator
Trauma Manufacturer Finger External Fixator
 Titanium Implants Distal Tibiofibular Set
Titanium Implants Distal Tibiofibular Set
 Titanium Alloy Spine Pedicle Screw Connector
Titanium Alloy Spine Pedicle Screw Connector
In the high-stakes realm of spinal surgery, structural stability is the cornerstone of successful clinical outcomes. Cross Link Connectors, also known as transverse connectors, serve as the mechanical bridge between bilateral longitudinal rods in posterior spinal fixation systems. Their primary function is to increase the torsional rigidity of the screw-rod construct, effectively preventing rotation and lateral shifting. As a leading China Cross Link Connector manufacturer, we recognize that these components are not mere accessories but vital instruments that ensure the long-term integrity of spinal fusions.
Global industrial status indicates an increasing reliance on these devices due to the rising prevalence of complex spinal deformities, such as severe scoliosis and kyphosis, which require robust stabilization. The industrial landscape is currently dominated by a shift toward biocompatible materials like Titanium Alloy (Ti6Al4V) and PEEK. Worldwide, the orthopedic implant market is projected to grow significantly as aging populations in North America, Europe, and Asia-Pacific demand higher quality of life through surgical intervention. For international procurement managers, sourcing from a reliable China Cross Link Connector exporter offers a strategic balance between cutting-edge engineering and cost-effectiveness.
Our flagship Spine Pedicle Screw Domino Connector represents the pinnacle of Chinese manufacturing precision. Designed for the 5.5mm rod system, this connector allows for seamless transition and multi-level stabilization. Utilizing aerospace-grade Titanium Alloy, it offers unparalleled strength-to-weight ratios and bio-inertness, ensuring that the patient's body accepts the implant without adverse reactions.
The ergonomic design facilitates easier intraoperative adjustments, reducing the surgeon's "fiddle factor" and shortening anesthesia time for the patient. For global distributors, this product meets the rigorous demands of modern trauma and degenerative spinal clinics.
Key Features: High Torsional Stability, Variable Angle Adjustability, CE Certified.
The orthopedic industry is undergoing a digital and biological revolution. One of the most prominent trends is the move toward minimally invasive surgery (MIS). Cross Link Connectors are now being redesigned to fit through smaller incisions, requiring low-profile shapes that do not irritate the surrounding soft tissue. Furthermore, the integration of 3D printing technology is allowing for patient-specific connectors that match the unique anatomical curvature of a patient’s spine.
Global procurement needs have shifted from simple commodity buying to "partnership-based sourcing." Hospitals and large medical groups are looking for suppliers who offer a comprehensive ecosystem: high-quality implants, specialized instrument sets, and responsive technical support. As a China Cross Link Connector manufacturer, we have adapted by providing full-service OEM/ODM solutions, ensuring our partners can brand high-quality hardware while we handle the complex manufacturing and regulatory compliance (MDR/FDA) processes.
Localization is another critical factor. In markets like Latin America and Southeast Asia, there is a massive demand for durable, affordable implants that can withstand the rigors of public healthcare systems. Our products are engineered to meet these local application scenarios, providing reliable performance in both high-end private hospitals and high-volume public trauma centers.
Beyond spinal connectors, our expertise extends to complex intramedullary fixation. The Titanium Pfna Proximal Femoral Intramedullary Nail is a testament to our diverse R&D capabilities. While Cross Link Connectors stabilize the axial skeleton, our PFNA systems stabilize the lower limbs, critical for geriatric patients suffering from osteoporosis-related hip fractures.
By leveraging the same high-grade titanium used in our spinal systems, these nails offer superior fatigue resistance. The synergy in our manufacturing process means that every implant, whether a small connector or a large femoral nail, undergoes the same 100% inspection protocol.
Application: Intertrochanteric fractures, high-strength fixation for elderly patients.
The "Made in China" label in the medical device sector now stands for "Quality at Scale." Our facility, founded in 1999, was among the pioneers of orthopedic R&D in China. Our advantages are manifold:
Founded in 1999, the company is one of the earliest enterprises in China engaged in the research and development of orthopedic implants, and has now become one of the leading companies in the industry, with products exported overseas. Sales channels cover more than 160 countries and regions. Local registration has been obtained in more than 60 countries and regions. More than 3 overseas offices have been established in countries such as the United States and the Netherlands. More than 100 global OEM partners. Currently, 214 products have obtained CE certification.
Our Legacy: Founded in 1999, we have dedicated over two decades to the mastery of orthopedic science. Our journey from a local R&D firm to a global leader is fueled by a commitment to patient safety and surgical precision.


The effectiveness of a Cross Link Connector is measured by its ability to resist torsional forces. During spinal fusion, the biological goal is to create a solid bone bridge between vertebrae. However, the human body is constantly subjected to rotational stresses during movement. Without a transverse connector, the two longitudinal rods may act independently, leading to "parallelogramming" or construct failure. Research shows that adding a Cross Link can increase torsional stiffness by up to 45% in multi-level constructs. This is particularly critical in the lumbar and thoracic regions where the range of motion is higher.
For the modern surgeon, the choice of connector involves balancing stability with "dead space." Larger connectors provide more stability but can increase the risk of infection or hardware prominence. Our R&D team has focused on a low-profile design that maintains high mechanical strength through superior alloy selection, ensuring that the implant remains flush with the spinal hardware, minimizing patient discomfort.
In trauma scenarios—such as burst fractures from high-velocity impact—the demand on the fixation system is extreme. Here, our Cross Link Connectors provide the necessary "frame" effect to keep the spinal column aligned during the critical healing phase. This reliability is why we are a preferred exporter for emergency trauma centers across the Middle East, South America, and Eastern Europe.
Furthermore, the environmental impact of medical manufacturing is an emerging concern. Our factory in China has implemented "Green Manufacturing" protocols, optimizing material usage and reducing waste in the CNC milling process. This commitment to sustainability is increasingly important for procurement tenders in the European Union and Northern European markets.
 Disposable Titanium Alloy Spine Pedicle Screw Connector
Disposable Titanium Alloy Spine Pedicle Screw Connector
 Premium Adult Shoulder Abduction Brace
Premium Adult Shoulder Abduction Brace
 Breathable Fabric Shoulder Sling Stability
Breathable Fabric Shoulder Sling Stability
 Adjustable Memory Foam Shoulder Pillow
Adjustable Memory Foam Shoulder Pillow
 Universal Shoulder Abduction Bracket
Universal Shoulder Abduction Bracket
 Ultimate Shoulder Abduction Brace for Recovery
Ultimate Shoulder Abduction Brace for Recovery
 Medical Orthopedic Titanium Implants Set
Medical Orthopedic Titanium Implants Set
 Titanium Pfna Proximal Femoral Intramedullary Nail
Titanium Pfna Proximal Femoral Intramedullary Nail