 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
 Titanium Orthopedic Femur Interlocking Nail Medical Bone Surgical Femoral Intramedullary Implant
Titanium Orthopedic Femur Interlocking Nail Medical Bone Surgical Femoral Intramedullary Implant
 Variable Angle Orthopedic LCP Distal Femoral Locking Plate
Variable Angle Orthopedic LCP Distal Femoral Locking Plate
 Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
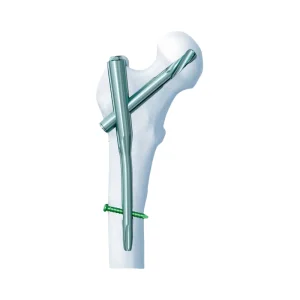 Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
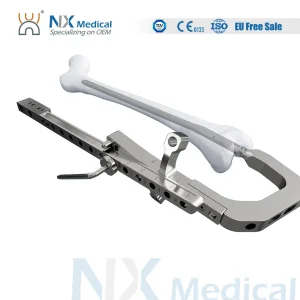 Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System
Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System
The global orthopedic market is witnessing a paradigm shift as the demand for high-performance Ceramic Femoral Heads continues to soar. As populations age and the need for durable hip replacements grows, medical institutions worldwide are moving away from traditional metal-on-metal or metal-on-polyethylene interfaces toward ceramic alternatives. Ceramic materials, such as Alumina-Zirconia composites, offer unparalleled hardness, bio-compatibility, and the lowest wear rates in the industry.
Today, Ceramic Femoral Heads are considered the "Gold Standard" for younger patients and those seeking a long-term solution (25+ years) for Total Hip Arthroplasty (THA). In Europe, North America, and the Asia-Pacific region, surgeons are increasingly specifying ceramic components to avoid the risks associated with metal ion release and "trunnionosis." As a leading manufacturer in China, we stand at the crossroads of this technological evolution, providing surgical-grade ceramics that rival global benchmarks in both safety and mechanical longevity.
Global Sales Channels in Countries & Regions
Local Registrations Obtained Globally
Products with CE Certification
Global OEM Strategic Partners
Our flagship Ceramic Femoral Head solutions are engineered using ultra-high-purity ceramics designed to minimize friction. In the procurement world, reliability is non-negotiable. Hospitals and distributors require implants that resist chipping and fragmentation. By utilizing advanced Hot Isostatic Pressing (HIP) technologies, we ensure our ceramic heads achieve a density and grain structure that provides superior fracture toughness.
This product line, including the Primary Total Hip System Hip Joint Replacement Surgery Femoral Head, is specifically designed to work harmoniously with our UHMWPE acetabular liners, reducing the volumetric wear by up to 90% compared to traditional metal-on-polyethylene systems. For international buyers, this means fewer revision surgeries and higher patient satisfaction rates.
Unlike cobalt-chromium heads, ceramics are inert. They do not release metallic ions into the bloodstream, making them the safest choice for patients with metal sensitivities.
Ceramic surfaces have higher "wettability," meaning they maintain a better film of synovial fluid, leading to smoother joint movement and less "squeaking."
As a vertically integrated manufacturer, we mitigate the risks of global shortages. We offer consistent lead times and competitive pricing without compromising on ISO/CE standards.
Global procurement trends indicate a shift toward high-volume, high-quality sourcing from certified Chinese factories. The "China-plus-one" strategy has led many medical device distributors to seek reliable OEM partners in China who can deliver the same quality as European Tier-1 brands but with significantly better logistics and cost-efficiency. Our role as a primary exporter fulfills this need, bridging the gap between high-end R&D and affordable healthcare accessibility.
Founded in 1999, our company was one of the earliest pioneers in the Chinese orthopedic implant industry. Over the past two decades, we have transformed from a local manufacturer into a global powerhouse. Our factory features ISO-certified cleanrooms and state-of-the-art CNC machining centers from Switzerland and Germany.
When you source from us, you aren't just buying a product; you are partnering with an entity that has 3+ overseas offices in the USA and Netherlands to provide localized support. Our Artificial Hip and Knee Joint Replacement series represents the culmination of 25 years of clinical data and iterative design. We maintain rigorous quality control checkpoints, from raw material inspection to the final sterilization packaging, ensuring every femoral head meets the highest safety criteria.
Whether it is a high-volume public hospital in Southeast Asia or a specialized private orthopedic clinic in Latin America, our Ceramic Femoral Heads are designed for versatile clinical application. They are compatible with both primary hip replacements and complex revision surgeries. For trauma cases, our integrated systems—including PFNA Intramedullary Nails and locking plates—ensure that surgeons have a comprehensive "toolbelt" for any orthopedic challenge.
Our Corporate Legacy:
Since our inception in 1999, we have focused on the R&D of orthopedic implants that change lives. Today, we are proud to have obtained local registration in more than 60 countries. This regulatory achievement is a testament to our quality management system. Our mission is to continue exporting "China's Precision" to the world, ensuring that high-quality surgical implants are available to all, regardless of geography.

Our manufacturing ecosystem is backed by leading medical groups, ensuring continuous innovation.
 Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System
Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System
 Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System
Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System
 CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery
CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery
 High Grade Surgical Orthopedic Instrument Set Knee Joint Femoral Instrument Set
High Grade Surgical Orthopedic Instrument Set Knee Joint Femoral Instrument Set
 Advanced Trochanteric Femoral Intramedullary Nail for Fracture Repair
Advanced Trochanteric Femoral Intramedullary Nail for Fracture Repair
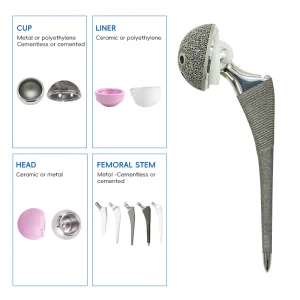 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System
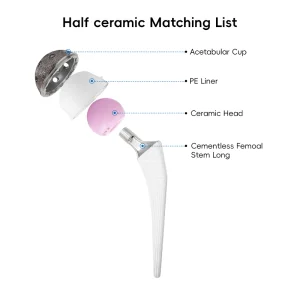 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
 Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China