 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants


Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery

Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery

Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar

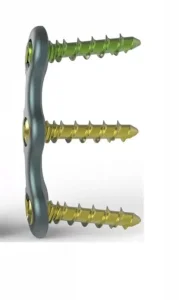
Anterior Cervical Fixation System with Screw for Cervical Spine with CE Certificate

Anterior Cervical Fixation System with Screw for Cervical Spine with CE Certificate

Medical Equipment: Cervical Pedicle Screw System with Locking Plate
The global market for Posterior Cervical Fixation Systems has witnessed exponential growth over the last decade. As the world population ages, the prevalence of degenerative spinal conditions such as cervical spondylotic myelopathy, disc herniation, and spinal stenosis has increased. Surgeons worldwide are increasingly opting for posterior stabilization techniques due to their robust biomechanical advantages in multi-level cervical pathologies.
Today, the industrial landscape is dominated by high-precision manufacturing. The shift from stainless steel to Titanium Alloy (Ti6Al4V) and PEEK (Polyetheretherketone) materials has revolutionized patient outcomes, offering better biocompatibility and MRI compatibility. The industrial status is no longer just about "hardware"; it is about the integration of advanced material science and ergonomic design that allows for easier implantation and faster patient recovery.
Mature healthcare systems in North America and Europe continue to drive demand for premium, multi-axial screw systems, while emerging markets in Asia and Latin America are seeing rapid adoption of these technologies.
Global orthopedic associations are standardizing surgical protocols, emphasizing the importance of posterior fixation in complex deformity corrections and trauma cases.
With global supply chains facing scrutiny, decentralized manufacturing and the rise of high-quality Chinese production centers have become vital for maintaining global healthcare stability.
As a leading Posterior Cervical Fixation System manufacturer, we keep a close eye on the technological horizon. The industry is moving toward "Smart Surgery" and "Biological Integration." Key trends include:
Note for Procurement Officers: The trend is moving away from generic sets toward procedure-specific kits. Our R&D team focuses on "low-profile" designs to minimize soft tissue irritation, which is a top priority for modern clinical evaluations.
When hospitals and distributors look for a Posterior Cervical Fixation System supplier, their requirements vary by region, but quality remains the universal language. In developed regions like North America and the EU, CE (MDR) and FDA certifications are non-negotiable. In Southeast Asia and Africa, there is a balanced demand for high-tier quality and cost-effectiveness.
1. Trauma & Emergency Care: In high-traffic urban centers, rapid-response spinal fixation is critical for stabilizing cervical fractures. Our systems provide the fast-locking mechanisms required for these high-pressure environments.
2. Degenerative Deformity Correction: In aging societies (like Japan, Korea, and parts of Europe), long-segment posterior fixation is often used to treat multi-level stenosis. The multi-axial capability of our pedicle screws allows surgeons to handle complex anatomical variations easily.
3. Oncology/Tumor Resection: When cervical vertebrae are removed due to tumors, our laminar hooks and rod connectors provide the necessary bridge to maintain spinal stability.
Founded in 1999, we are pioneers in the Chinese orthopedic industry. Our journey from a local R&D lab to a global powerhouse is built on the foundation of precision and trust. Here is why we stand out as the best posterior cervical fixation system manufacturer in China:
Our facility utilizes the latest German and Swiss CNC machining centers to ensure tolerances within microns. Every screw and plate is a masterpiece of engineering.
With 214 CE certificates and registrations in over 60 countries, we understand the regulatory hurdles of the international medical market.
We serve over 100 global OEM partners, providing customized solutions that meet specific market needs, from branding to specialized instrument sets.
Our location in China allows us to leverage a massive supply chain for raw materials like Grade 5 Titanium while maintaining strict quality control. This allows us to offer pricing that enables hospitals to provide high-quality care without the "brand premium" associated with traditional Western giants.
Established in 1999, we are one of the earliest enterprises in China dedicated to the R&D of orthopedic implants. With overseas offices in the United States and Netherlands, we provide 24/7 support to our global clientele.



Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation

High Quality Cannulated Axial-Rotation Pedicle Screw Orthopaedic Implant

Cannulated Axial-Rotation Pedicle Screw Surgical Instrument System

Titanium Alloy Spine Pedicle Screw Domino Connector Surgical Instrument

Disposable Titanium Alloy Spine Pedicle Screw Domino Connector 5.5 System

Sacrual Self-Breaking Plug Titanium Pedicle Screw Orthopaedic Implant

Peek Cervical Interbody Fusion Cage with 2 Locking Screws, Titanium Alloy

Wholesale High Quality Titanium Coating Surgical Instrument Set