 Blutree Orthopedic Implants
Blutree Orthopedic Implants
 Blutree Orthopedic Implants
Blutree Orthopedic Implants
The global bone fixation device market is currently undergoing a significant transformation. As the global population ages, the incidence of osteoporosis-related fractures and trauma cases has surged, driving the demand for high-reliability fixation systems. From traditional stainless steel plates to advanced titanium alloys and bio-absorbable materials, the industry is shifting toward solutions that promote faster healing and minimize secondary surgeries.
Modern bone fixation is no longer just about mechanical stability. The integration of 3D printing (additive manufacturing) allows for patient-specific implants that match the unique anatomy of complex fractures. As a leading China Bone Fixation Device Factory, we are at the forefront of combining biomechanical engineering with computer-aided design to produce devices that reduce stress shielding and enhance osseointegration.
Hospitals and healthcare providers worldwide are facing mounting pressure to reduce costs without compromising patient outcomes. This has led to a significant shift in procurement strategies, where high-quality medical devices from China are increasingly preferred over expensive Western brands. The ability to provide CE-certified orthopedic implants at a competitive price point is the hallmark of the current industrial era.
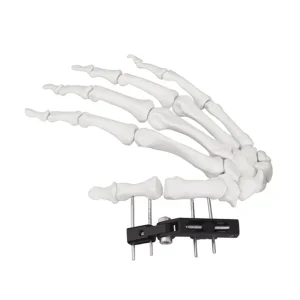
Our External Fixator Hand Bone Pin Stent is engineered for distal radius fractures, providing superior stability through a track vertical axis design. This device allows for precise adjustment and micro-compression, which is vital for maintaining joint alignment during the critical healing phase.
Link to Product: External Fixator Hand Bone Pin Stent
The future of orthopedic bone fixation is defined by "Intelligence" and "Miniaturization". We are seeing a move toward smart implants that can monitor healing progress through embedded sensors. While still in early stages, the groundwork is being laid in the precision manufacturing of the screw-plate interface to prevent loosening.
Another major trend is the development of minimally invasive surgical (MIS) instruments. Surgeons now prefer fixation devices that can be implanted through smaller incisions, reducing blood loss and recovery time. Our R&D team focuses on developing cannulated screws and low-profile plates that accommodate these modern surgical techniques.
Global distributors and hospital groups are looking for more than just a product; they are looking for a reliable supply chain partner. In the current geopolitical and economic climate, stability of supply and regulatory compliance are the top priorities for international buyers.
Accessing markets in the EU, USA, and Southeast Asia requires rigorous certification. We provide full documentation including CE, ISO 13485, and local registrations to ensure seamless entry into your local healthcare system.
Many global enterprises require customized branding or modified designs to suit specific surgical habits in their region. Our factory offers comprehensive OEM support, from CAD drawing to final sterile packaging.
Medical emergencies don't wait. We maintain a high inventory level of standard sizes for spinal screws, trauma plates, and external fixators to ensure rapid global dispatch within 72 hours for urgent orders.

Our Bone Cement-Injectable Cannulated Pedicle Screws represent the pinnacle of spinal surgery technology. Designed for patients with poor bone quality (osteoporosis), these screws allow for the controlled delivery of bone cement to enhance pull-out strength and stability.
Link to Product: Factory Direct Spinal Implant
Bone fixation devices are utilized across a spectrum of medical environments, each with unique requirements:
In emergency high-energy trauma (car accidents, falls), our External Fixation Systems are the first line of defense. They provide rapid stabilization of open fractures before definitive internal fixation can be performed.
For hip and radius fractures in the elderly, our Locking Compression Plates (LCP) are essential. The angular stability provided by the locking mechanism ensures that screws do not back out of osteoporotic bone.
Degenerative disc diseases and scoliosis correction require high-precision Pedicle Screw Systems. Our systems provide the rigid fixation needed for successful spinal fusion and deformity correction.
Founded in 1999, we have spent over two decades perfecting the art of orthopedic manufacturing.
As a premier Bone Fixation Device Manufacturer in China, our advantage lies in the perfect balance of scale and precision. We operate state-of-the-art CNC machining centers from Switzerland and Japan, ensuring that every screw thread and every plate contour meets micron-level tolerances.
Our R&D department collaborates with top surgeons to refine our products based on real-world clinical feedback. This iterative process has allowed us to establish over 100 global OEM partnerships and set up dedicated offices in the United States and the Netherlands to provide localized support for our Western clients.
Keywords: Bone Fixation Device Manufacturer, Orthopedic Implants Factory China, External Fixator Supplier, Spinal Pedicle Screw Production, Titanium Bone Screws, Trauma Fixation Implants, CE Certified Medical Devices, OEM Orthopedic Implants, Bone Fracture Surgery Solutions.